9_Application_News_No_SCA_210_051
Liquid Chromat ography Mass Spectr ometr y Analysis of thiaminpyrophosphat e and pyridox al - 5’ - phosphate in whole blood using fully automated sample preparation LC/MS/MS system (CLAM- 2000 + LCMS - 8045) Introduct …

Liquid Chromatography Mass Spectrometry
Analysis of thiaminpyrophosphate and pyridoxal-5’-
phosphate in whole blood using fully automated sample
preparation LC/MS/MS system
(CLAM-2000 + LCMS-8045)
Introduction
Vitamin B1, thiamin, plays an important role in the
metabolic pathway in the human body. The
biological active form is Thiamin Pyrophosphate
(TPP). The water soluble vitamin acts as a
coenzyme for the enzymatic degradation of
glucose in the citric acid cycle. A non-varied diet or
malnutrition can quickly lead to a deficiency which
can result in inter alia depression, muscle
weakness and tachycardia. Vitamin B6 has
multiple forms. The biological active form in the
human cell is Pyridoxal-5’-Phosphate (P5P). The
water soluble vitamin acts as a coenzyme in the
formation of amino acids, amines and peptides. In
case of a deficiency the other B vitamins will also
be deficient. A PLP deficiency can occur due to
chemotherapy, alcoholism, pregnancy and kidney
failure.
These two vitamins are predominately analysed
with HPLC and fluorescence detection, which
requires derivatization. More recent there is a lot of
development for the analyses of these vitamins
with LC-MS/MS and protein precipitation. Due to
rising number off samples an automated solution is
needed. The aim of these study was to set-up an
automated solution for the analysis of TPP and
P5P in whole blood, using the kit from
Instruchemie in combination with the CLAM-2000
and LCMS-8045.
Materials and Methods
The quantitative analysis of TPP and P5P from
whole blood samples was performed using reagent
provided in Instruchemie Vitamine B1&B6 kit.
Chromatography separation was developed using
Mobile Phase 1 (ref 3156) and Mobile Phase 2 (ref
3157) and a Shimpack GIST C18-AQ column (ref
227-30765-03). Both vitamins and their internal
standards were monitored using a LC-MS/MS
system (Nexera X2 and LCMS-8045, Shimadzu,
Kyoto) following specific MRM transitions.
Sample preparation was performed using
precipitation reagent (ref 3169) and internal
standard mix (ref 3153). Analytical performance of
the method was monitored using whole blood
calibrators (ref 3168) and whole blood quality
control samples (ref 3159). Automatic sample
preparation was performed using CLAM-2000
(Shimadzu, Kyoto).
No. SCA_210_051
Figure 1: Fully automated Sample Preparation
LC-MS/MS system
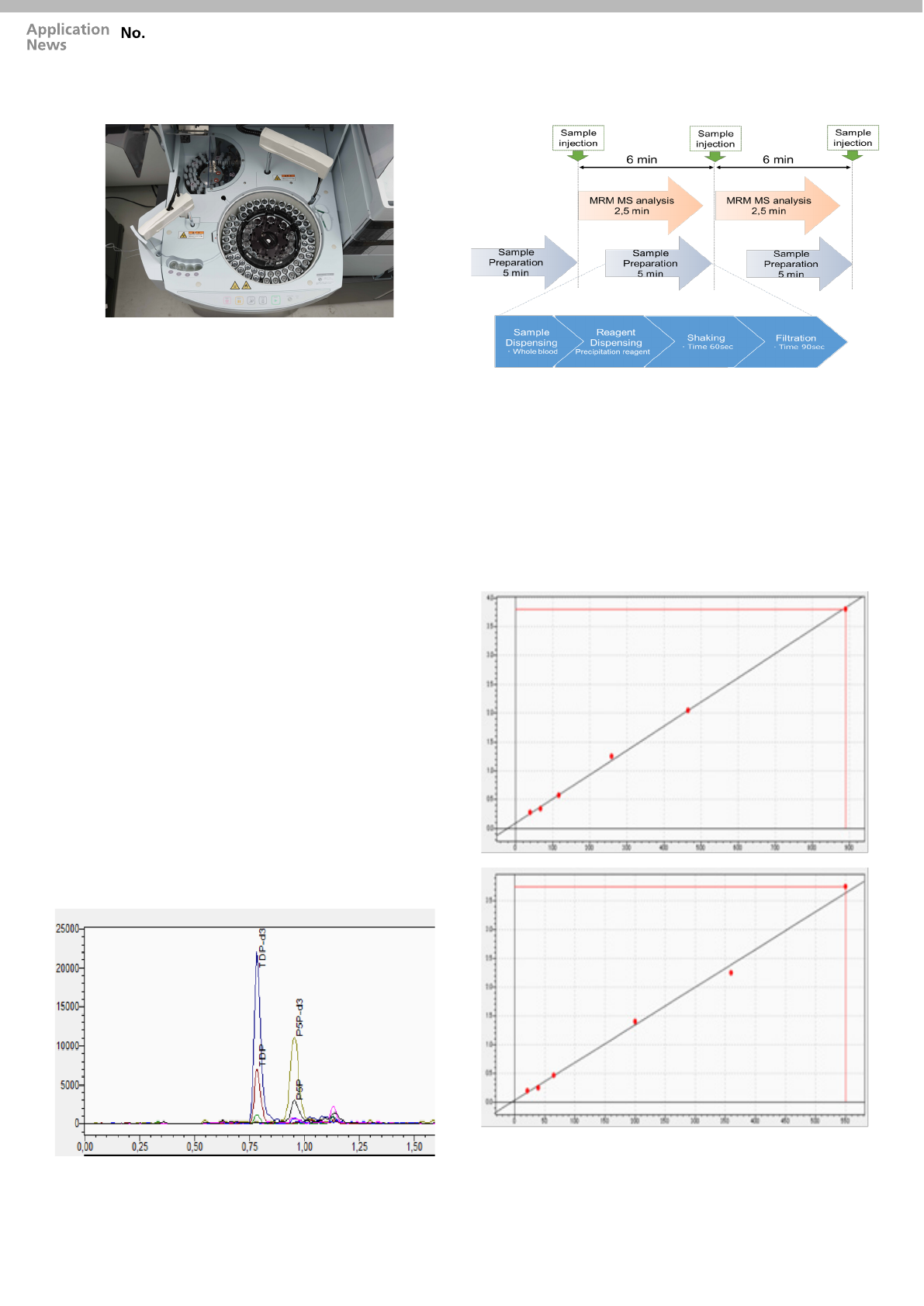
Fully automated sample preparation
With the aim to reduce the operator involvement,
to increase the throughput and data quality, the
manual sample preparation procedure was
substituted by an automatic procedure using a
novel Clinical Laboratory Automated sample
preparation Module (CLAM-2000) online with the
LC-MS/MS system (LCMS-8045).
Blood collection tubes were loaded together with
disposable micro-vial containing calibrators and
quality control samples. Precipitation reagent and
ISTD mix are placed into specific instrument slot
(8 ºC). The fully automated sample preparation
procedure contains all steps from sample
collection to LC-MS/MS analysis (Figure 2).
Results
Figure 3 shows a representative mass
chromatogram of TPP and P5P and their internals
standards from a control sample at low level.
Calibration curves were prepared by continuous
analysis with fully automated sample preparation
and analysis and used to asses accuracy and
precision. Good linearity was obtained across the
set calibration range for both vitamins, with
accuracy of 100 ± 15% over the entire
measurement range including limit of
quantification.
SCA_210_051
Figure 2: Schematic preparation/analysis workflow
Figure 3: Low control sample TPP/P5P 49/31 nmol/L
TPP y=0.00420734x + 0.0902752
r
2
= 0.9973214
P5P y=0.00656861x + 0.0295692
r
2
= 0.9954683
Figure 4: Calibration curves for TPP (39 – 890
nmol/L) and P5P (21 – 550 nmol/L)
© Shimadzu Europa GmbH 2018
www.shimadzu.eu
Shimadzu Europa GmbH
Precision was measured at a % RSD of within
15% showing that good reproducibility was
achieved. These results indicate that automated
sample preparation and analysis with the
CLAM-2000-LCMS-8045 system is suitable for the
analysis of the biological active forms of vitamin
B1 and B6 in whole blood samples.
Conclusion
Results indicate that the fully automated sample
preparation LC/MS/MS system in combination with
the Instruchemie vitamine B1&B6 kit can eliminate
the risk of error of variability introduced by manual
sample preparation which is often a problem for
whole blood analysis. Also the results indicate that
the quick and high precision analytical workflow
from the CLAM-2000 system in combination with
the LCMS-8045 is a perfect solution for the high
amount of whole blood samples that are analysed
for TDP and PLP in the clinical chemistry.
SCA_210_051