IPC-TM-650 EN 2022 试验方法-- - 第177页
extraction solution before measuring the resistivity of the com- posite sa mple. 4.2 Tes t Equipment and Chemicals 4.2.1 Miscellaneous laboratory ware (e.g., beakers, funnel s, storage bottles an d graduated cy linders).…

1 Scope
1.1 Purpose
These tests are used as process control
tools; they can be used to inspect printed boards or printed
board assemblies and determine if they conform to the moni-
toring level of the user’s performance specification. Bulk ionic
cleanliness testing may be accomplished by measuring the
ionizable surface contaminants extracted by the following
three methods:
1. Manual extraction method
2. Dynamic extraction method
3. Static extraction method
Please note that this method does not predict reliabil-
ity and should only be used as a process control tool.
1.2 Restrictions
Measurements of ionic conductivities do
not differentiate between different ionic species. They simply
measure conductivities (or resistivities) which can be related to
amounts of ionic materials present in solution. There is no
identification of the contribution to the total conductivity read-
ings of any individual ionic species which may be extracted
into the solution. For measurement of individual ionic species
(type and level of residue) see IPC-TM-650, Test Method
2.3.28, Ionic Analysis of Circuit Boards, Ion Chromatography
Method.
For simplicity, amounts of ionic materials in solution can be
expressed by a conductivity factor which is equivalent to the
measured conductivity contributed by a known amount of a
standard, strongly ionized salt such as sodium chloride (NaCl).
Ionic residues are therefore usually expressed as equivalents
of sodium chloride in micrograms per unit surface area (e.g.,
µg NaCl Eq./cm
2
) of the sample. This does not imply that the
contamination is NaCl but, rather, it exhibits a conductivity
function which is equivalent to that of the expressed amount
of sodium chloride if it were in solution instead of the ionic soil.
These tests will not measure any surface ionic materials which
are not brought into solution because of insolubility, physical
entrapment or inadequate exposure to the extracting solvent.
Additionally, non-ionic components of the soil will not be mea-
sured.
1.3 Application
Caution should be exercised in comparing
results between different test equipment as well as the differ-
ent test methods. These methods are applicable as quality
control tools in evaluating the parameters of materials and
cleaning process, in terms of how they affect the final cleanli-
ness of the board or assembly. As process control tools, they
can be used to inspect printed wiring boards or printed wiring
assemblies and determine if they conform to the requirements
of the user’s performance specification. It is important to
understand, no calculation factor can exist to convert data
from one style of commercial tester to another due to the
great differences in system responses. See IPC-TR-583. This
means it is very difficult to compare data from different
machines. These procedures can also be used to assist
in-process development as general residue indicators, to
evaluate flux cleanability, solvent efficiency and general
improvements of process parameters.
Please note that this method does not predict reliabil-
ity and should ONLY be used as a process control tool.
2 Applicable Documents
An In-Depth Look at Ionic Cleanliness Testing
3 Test Specimen
Follow the equipment manufacturer’s
recommendations as to sample size. Recommended area cal-
culations for the unpopulated printed board and printed board
assembly are the following:
Printed Board Surface Area = Length x Width x 2
Printed Board Assembly Surface Area = (Length x Width x 2)
+ (1 x up to 50% of the board area)
Report the surface area used in the calculation and the per-
centage increased in calculating the surface area of a printed
board assembly.
There is no universally accepted method for determin-
ing the surface area of components. However, the determina-
tion of surface area for components should be established ini-
tially and used whenever that assembly style is being tested.
4 Manual Extraction Method
4.1 Description
This method describes the manual extrac-
tion of ionic material from the surface of a sample. This con-
sists of flushing the board surface with a stream of
2-propanol/DI water mixture and carefully capturing all of the
3000 Lakeside Drive, Suite 309S
Bannockburn, IL 60015-1249
IPC-TM-650
TEST METHODS MANUAL
Number
2.3.25
Subject
Detection and Measurement of Ionizable Surface
Contaminants by Resistivity of Solvent Extract (ROSE)
Date
11/12
Revision
D
Originating Task Group
Ionic Conductivity / Ion Chromatography Task Group
(5-32a)
Association
Connecting
Electronics
Industries
Note:
Note:
IPC-TR-583
Note:
Material
/n
this
Test
Methods
Manual
was
voluntarily
established
by
Technical
Committees
of
I
PC.
This
material
/s
advisory
only
and
"s
use
or
adaptation
,
s
entirely
voluntary.
IPC
disclaims
all
liability
of
any
kind
as
to
the
use,
application,
or
adaptation
of
this
material.
Users
are
also
wholly
responsible
for
protecting
themselves
against
all
claims
or
liabilities
for
patent
infringement.
Equipment
referenced
/s
for
the
convenience
of
the
user
and
does
not
imply
endorsement
by
IPC.
Page
1
of
5

extraction solution before measuring the resistivity of the com-
posite sample.
4.2 Test Equipment and Chemicals
4.2.1
Miscellaneous laboratory ware (e.g., beakers, funnels,
storage bottles and graduated cylinders). This plastic ware
can be high density polyethylene, polymethylpentene (poly-
pentene), polypropylene or equivalent. Glassware cannot be
used because it has been shown to contribute ionic contami-
nation in a short time with this 2-propanol/DI water solution.
4.2.2
Conductivity bridge or equivalent conductivity probe
and temperature compensated liquid conductivity cell appara-
tus capable of measuring specific resistivities within a range
covering at least 100 kΩ-cm to 20 MΩ-cm.
4.2.3
Deionization column. Up to 100% solvent compatible
mixed bed or equivalent.
Some of these columns are color dyed. This dye will
interfere with test results. Make certain that the column used
has no dye.
4.2.4
Electronic Grade 2-Propanol (isopropyl alcohol) or
99.5% purity minimum
4.2.5
Deionized water (DI water), preferably above 16
MΩ-cm water resistivity
It is critical to always use wash solution with the same
composition of 2-propanol/DI water for all comparative data
discussions.
4.2.6
Wash solution composed of 75 ± 2% v/v 2-propanol/
DI water or 50 ± 2% v/v 2-propanol/DI water. This wash solu-
tion must be deionized to a resistance equal to or greater than
16 MΩ-cm (conductivity less than 0.0625 µS/cm). If stored,
this wash solution must be freshly deionized prior to use. Typi-
cal resistivity of 25 MΩ-cm (conductivity of 0.04 µS/cm) can
be achieved. Measurement of the 2-propanol/DI water solu-
tion should be performed using a calibrated hydrometer. The
default extract solution composition is 75 ± 2% v/v
2-propanol/DI water.
4.3 Calibration of Bridge
This is essential in the manual
method because there can be no correlation between
resistivity/conductivity readings and NaCl equivalents without
calibration. All future specification requirements are to be
stated in maximum micrograms of NaCl equivalent per square
centimeter (µg NaCl Eq./cm
2
).
Ensure that a proper cell constant value is considered
in any conductivity calculations. Check the specific conductiv-
ity bridge manufacturer’s manual for this detail.
4.3.1
Prepare or purchase (these standard solutions are
available from equipment vendors, as well as many of the
chemical companies) a standard NaCl solution from a weight
of ACS reagent grade NaCl salt dissolved in deionized water
(16 MΩ-cm resistivity minimum) to produce a final diluted con-
centration of 0.06 g/liter NaCl (5 mL contains 300 µg NaCl).
It is recommended that dry NaCl be used for this solu-
tion. A recommended drying exposure is one hour at 105 °C
or higher.
4.3.2
Place one liter of the 2-propanol/DI water solution (at
the calibration temperature of the bridge in use) in a plastic
beaker.
The 75%/25% v/v or 50%/50% v/v 2-propanol/DI
water solution must be used in this calibration. Water cannot
be used since it is not the test solution used in the procedure.
The test solution used in this calibration should be made fresh
daily.
4.3.3
From a 50 mL burette, add to the liter of test solution,
5 mL of the standard 0.06 g/liter NaCl solution. Stir and mea-
sure resistivity/conductivity.
If the resistivity is measured, convert to conductivity by
take the reciprocal, prior to plotting the data.
4.3.4
From a 50 mL burette, add to the liter of test solution,
20 additional mL of the standard 0.06 g/liter NaCl solution, for
a total of 25 mL. Stir and measure resistivity/conductivity.
4.3.5
From a 50 mL burette, add to the liter of test solution,
25 additional mL of the standard 0.06 g/liter NaCl solution, for
a total of 50 mL. Stir and measure resistivity/conductivity.
4.3.6
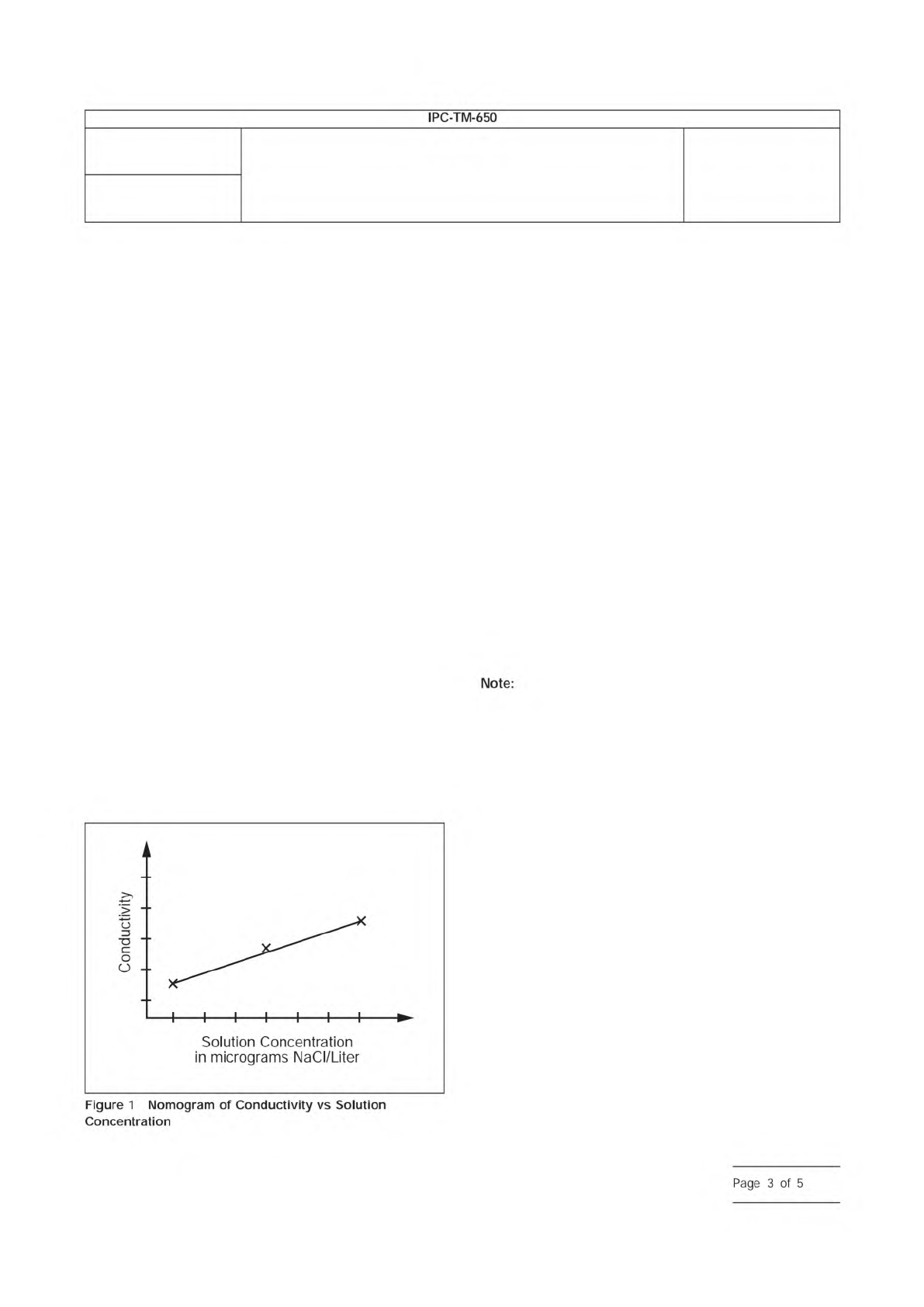
Plot a three-point nomogram of Conductivity vs.
µg/liter of NaCl. A linear relationship is expected with the use
of a linear regression best fit line through the data points. If a
multi-range meter is used the curve should not be extended
beyond the maximum reading of the meter for that range,
unless linearity is proven by additional points obtained by add-
ing more standard salt solution. The nomogram will never
cross the zero point of conductivity.
An R
2
value of 0.99 should be expected before the
instrument would be considered calibrated.
Number
2.3.25
Subject
Detection and Measurement of Ionizable Surface Contaminants by
Resistivity of Solvent Extract (ROSE)
Date
11/12
Revision
D
IPC-TM-650
—
Note:
Note:
Note:
Note:
Note:
Note:
Note:
Page
2
of
5
4.4 Test Procedure
4.4.1
Carefully preclean all plasticware with deionized water
(16 MΩ-cm resistivity minimum) followed by a final rinse with
the extraction test solution.
4.4.2
Determine the surface area per Section 3.
4.4.3
Suspend the test specimen within an appropriately
sized funnel positioned over a graduated cylinder. Use clean
gloves when handling the samples to be tested.
4.4.4
Prepare the extraction solution volume using a general
ratio of no more than 10 mL: 1 cm
2
of area. Direct a fine
stream of freshly-deionized test solution on both sides of the
specimen, covering all board and component surfaces. Con-
tinue this process, slowly collecting the extraction solution.
The volume collected is not critical, but the total collected vol-
ume must be exactly recorded. A volume correction is made
in the calculation.
4.4.5
Pour the final measured volume into a plastic ware
beaker, stir and measure the resistivity/conductivity with either
a bridge probe or equivalent conductivity probe.
4.4.6
The resistivity/conductivity readings can be used to
convert the µg NaCl equivalent as follows:
1. Locate where the resistivity or conductivity intersects the
calibration curve on the X-axis (see Figure 1).
2. Extend a vertical line from the point of intersection to the
x-axis. Read and record µg/liter NaCl (M).
3. Multiply the concentration in µg/liter NaCl by the total liters
of test solution used (V). This result indicates the total µg of
NaCl equivalents removed from the printed wiring board
(T).
T = M x V
Where:
T = the numerical value of the total amount of NaCl equiva-
lents removed from the printed board, expressed in micro-
gram (µg);
M = the numerical value of the NaCl concentration of test
solution, expressed in microgram per liter (µg/L);
V = the numerical value of the total volume of test solution,
expressed in liter (L)
4. Divide the micrograms of NaCl equivalents by the area of
the printed wiring board or assembly (A). This yields the
conductivity factor in µg NaCl Eq./cm
2
.
T/A = µg NaCl Eq./cm
2
If samples read over the highest standard the sample
should be diluted by a known factor, retested and calculations
adjusted accordingly.
5 Dynamic Extraction Method
5.1 Description
In the dynamic method, a purified
2-propanol/DI water mixture is circulated into and out of a test
tank chamber containing the sample being tested. The mix-
ture exiting the test tank is passed through a conductivity cell
which measures the conductivity continuously. These conduc-
tivity values are integrated over the time of the extraction. The
mixture is then pumped through a resin deionization column
before it is recirculated back into the test tank. As ionic mate-
rials are extracted from the samples and then pumped out of
the cell, the conductivity of the solution will change dynami-
cally until all of the extractable ionic material has been
removed.
5.2 Test Equipment
Dynamic conductivity measurement
system includes a test tank, a temperature compensated con-
ductivity cell, ion exchange columns and a metering pump
connected together in a recirculating loop as described in 5.1.
The conductivity readings are integrated over the time of the
measurement by electronic integration. The equipment may
have the capability of heating the 2-propanol/DI water mixture
IPC-2325d-1
Number
2.3.25
Subject
Detection and Measurement of Ionizable Surface Contaminants by
Resistivity of Solvent Extract (ROSE)
Date
11/12
Revision
D
IPC-TM-650
Solution
Concentration
in
micrograms
NaCI/Liter
Note:
Figure
1
Nomogram
of
Conductivity
vs
Solution
Concentration
Page
3
of
5