5_PO-CON1693E - 第3页
3 Fully automated platform for determination of immunosuppressant drugs in whole blood The Immunosuppressant standards and the Internal were rstly analysed by ow injection to optimize mass spectrometer parameters. All …
2
Fully automated platform for determination of
immunosuppressant drugs in whole blood
Introduction
Therapeutic drug monitoring of four major
immunosuppressant drugs, Cyclosporine A, Tacrolimus,
Sirolimus and Everolimus, is well established. Overdosing
with these critical dose drugs can cause serious toxicity
and long term morbidity, while organ rejection can occur
if a patient is under dosed. Nowadays clinical laboratory
has two main choices in technologies: immunoassay or
chromatography based methods. LC–MS/MS's superior
specicity makes it the presumptive gold standard in
immunosuppressant quantitation. It relieves the method
from common interferences that plague immunoassays
such as metabolites that have structural resemblance and
interfering antibodies. However, current LC–MS/MS
platforms demand personnel expertise and tedious
sample preparation and sample throughput is generally
much lower compared to immunoassays. We report a
fully automated procedure for the quantitation of four
major immunosuppressant in whole blood samples,
increasing data quality/precision, throughput and safety
(The work described herein is for research use only).
Methods
The quantitative analysis of Immunosuppressant was
performed using reagents provided in Chromsystems
“MassTox
®
” ONEMinute Kit (ref, 93900). The
Immunosuppressant and Internal standard were
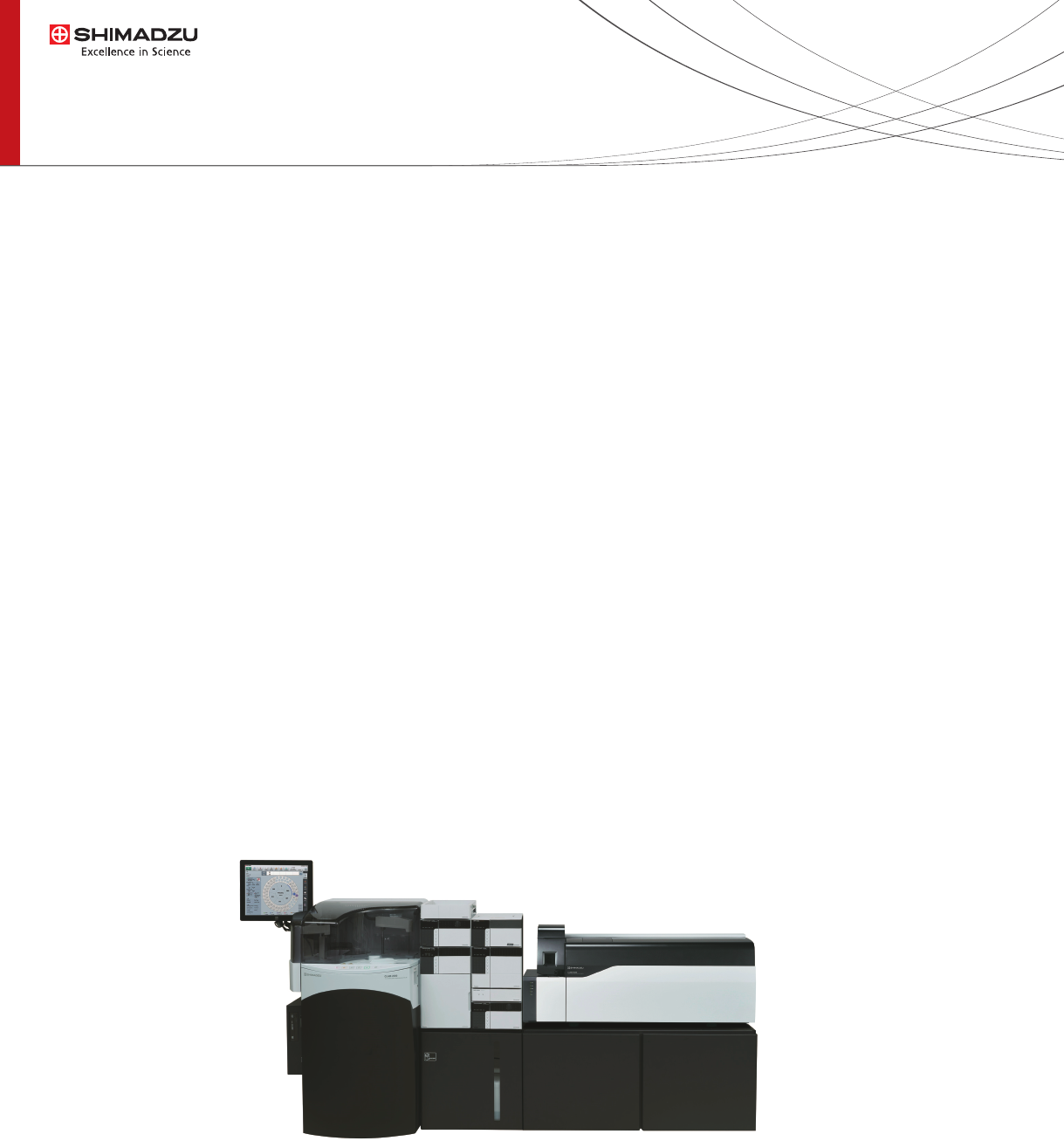
monitored using UHPLC-MS/MS system (Nexera X2 and
LCMS-8050, Shimadzu, Kyoto, Figure 1)).
Sample preparation was performed using Precipitation
reagent, Extraction buffer and Internal standard set.
Analytical performance of the method was monitored
using whole blood calibrators and whole blood QC.
Automatic sample preparation was performed using
CLAM-2000 module (Shimadzu, Kyoto) Figure 1.
Figure 1: CLAM-2000 online with Nexera X2 system and LCMS-8050 triple quadrupole mass spectrometer.

3
Fully automated platform for determination of
immunosuppressant drugs in whole blood
The Immunosuppressant standards and the Internal were rstly analysed by ow injection to optimize mass
spectrometer parameters. All compounds were detected as positive ion choosing the MRM transitions listed in Table 2.
Then Immunosuppressant standard mix was used to set-up chromatographic separation (Table 1).
LC-MS/MS analysis
Result and discussion
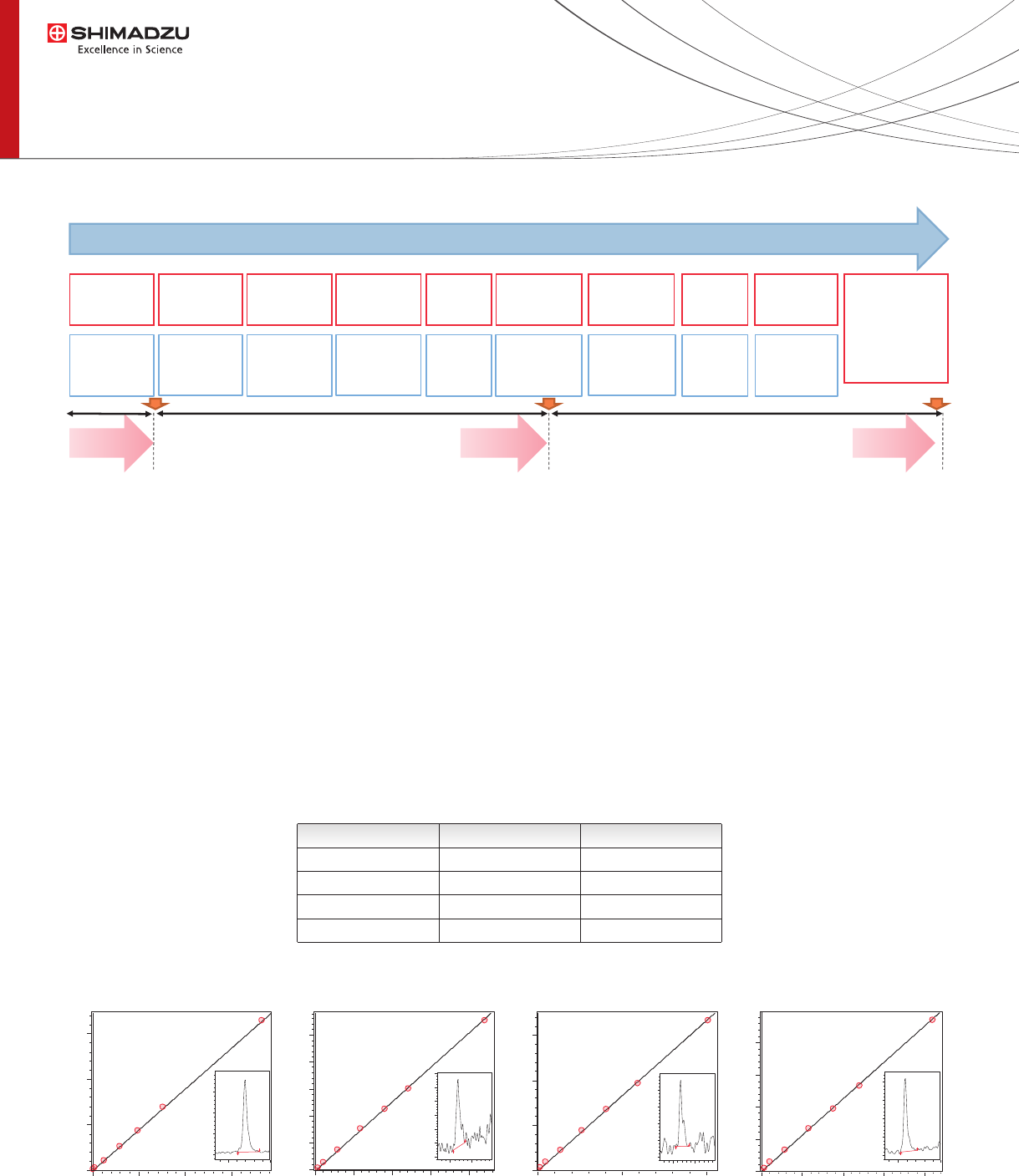
The CLAM-2000 was programmed to perform sample extraction and protein precipitation followed by ltration and
sample collection. The ltrated sample was then automatically transported using an arm from the CLAM-2000 to the
HPLC for LC-MS/MS analysis and no human intervention was required (Fig 2).
Fully Automated sample preparation
Column Temp. : 65 °C
Time Program : 0.3 min (trap load); 1.5 min (elution); 2.3 min (stop)
Injection Volume : 5 μL
[LC] NexeraX2 System
Ionization : ESI Positive
Nebulizer Gas : 3 L/min
Interface temperature : 300 °C
Desolvation Line : 250 °C
Heat Block temperature : 400 °C
Drying Gas : 10 L/min
Scan Type : MRM
[MS] LCMS-8050
Table 1: Analytical Condition
Table 2: MRM Transitions
Cyclosporin A
Tacrolimus
Sirolimus
Everolimus
Cyclosporin A-d
12
Everolimus-d
4
Sirolimus-d
3
Tacolimus-
13
Cd
2
Compound
1219.90 > 1202.80
821.60 > 768.30
931.70 > 864.50
975.70 > 908.50
1231.90 > 1214.80
979.60 > 912.40
934.60 > 864.40
824.60 > 771.40
MRM transition
4
Fully automated platform for determination of
immunosuppressant drugs in whole blood
Figure 3: Linearity and LLOQ.
Figure 2: CLAM2000 fully automated sample preparation and analysis.
Due to the overlapped sample preparation the throughput of the instrument was
1 result each 3.7 minutes for Immunosuppressant quantication.
Linearity and Accuracy were evaluated using reference
whole blood calibrators (7 levels) spanning from a wide
range of concentrations (Cyclosporin 16.5 – 1100 µg/mL,
Everolimus 0.86 – 32.7 µg/mL , Sirolimus 0.9 – 32.7
µg/mL, Tacrolimus 0.96 – 34.8 µg/mL). For all analytes
Linearity was >0.99 (see Table 3) with S/N >25 for LLOQ
levels (Figure 3). Precision of the assay was evaluated
using Chromsystems reference materials (whole blood
controls levels) spanning from low concentration to
highest concentration for each analyte (Table 4).
Linearity, Accuracy and Precision
Reagent
Dispensing
Extr. Buffer
50 ul
Sample
Dispensing
Whole blood
25 ul
Internal STD
12.5 ul
Reagent
Dispensing
Stirring
2200 rpm
Incubation
Room
temperature
Reagent
Dispensing
Filtration
Aspiration
Direct UHPLC
connection for
LCMS analysis
(2.3 min)
Stirring
2200 rpm
Parallel/Sequential sample preparation and analysis
Reagent
Dispensing
MetOH
20 ul
Sample 3
processing
Sample 2
processing
Sample 1
result
Prec. Reagent
125 ul
Table 3: Linearity & Accuracy.
Cyclosporin A
Tacrolimus
Sirolimus
Everolimus
Compound
90.4%-103.6%
99.7%-101-3%
95.1%-105.5%
96.9%-107%
Accuracy
0.998
0.997
0.998
0.998
r
2
0 500 1000 Conc. Ratio
0,0
2,5
5,0
7,5
Area Ratio
1,5 2,0
0
5000
10000
15000
0 10 20 30 Conc. Ratio
0,0
0,5
1,0
1,5
2,0
2,5
Area Ratio
1,50 1,75
0
250
500
750
1000
1250
1500
0,0 25,0 Conc. Ratio
0
1
2
3
Area Ratio
1,50 1,75
0
500
1000
1500
2000
0 10 20 Conc. Ratio
0,0
0,5
1,0
1,5
2,0
Area Ratio
1,5 2,0
0
2500
5000
7500
10000
Cyclosporin A Everolimus Sirolimus Tacrolimus