6_PO-CON1602E - 第3页
3 High throughput analysis for novel oral anticoagulants using LC-MS/MS system Integrated with automated sample preparation System Column : Mastro C18 2.1*100mm, 3μm Column Temp. : 50 ºC Mobile Phase A : 0.1% Formic Acid…

2
High throughput analysis for novel oral anticoagulants using
LC-MS/MS system Integrated with automated sample preparation
Introduction
LC-MS/MS has become an essential tool for monitoring
the concentration of drugs in biological samples due to its
high level of sensitivity and specicity; however, manual
sample preparation often involves several complicated
manual steps which can introduce error into the results.
Additionally, the time consuming nature of the sample
preparation and the large number of samples makes
LC-MS/MS a less desirable method. Automated sample
preparation has been shown to eliminate human error, as
well as increase laboratory efciency, making LC-MS/MS a
feasible method to incorporate into workows requiring
high throughput.
In this study, we investigated the ability to analyze for
Novel Oral Anticoagulants (NOACs) by LC-MS/MS
(LCMS-8040, Shimadzu) using automated sample
preparation (CLAM-2000, Shimadzu) to process large
sample sets. The CLAM-2000 has the ability to perform a
variety of steps appropriate for automated sample
preparation by LC-MS/MS including precipitation,
ltration, heating, shaking, and pipetting. This system is
seamlessly integrated with the LCMS system requiring no
human involvement after loading the biological samples
into the sample chamber. We validated the automated
method by comparing the data collected on the
automated system to the data collected using a manual
sample preparation protocol. An additional consideration
we evaluated was the number of human hours dedicated
towards preparing samples for this assay using both
methods and the ability to incorporate a STAT sample
into the queue with rapid turn-around time for results.
Method
Plasma or plasma spiked with NOACs was loaded directly
into the CLAM-2000 for sample processing. The
CLAM-2000 was programmed to perform protein
precipitation using acetonitrile followed by ltration and
sample collection. The sample was then transported using
an arm from the CLAM-2000 to the HPLC for LC-MS/MS
analysis and no human intervention was required. For the
manual sample preparation, acetonitrile precipitation
followed by centrifugation was performed. After the
sample preparation the automated and manually
prepared samples were analyzed using the same
LC-MS/MS method.
Instruments and LC-MS/MS analytical condition
Fig.1 CLAM-2000 and LCMS-8040 system
3
High throughput analysis for novel oral anticoagulants using
LC-MS/MS system Integrated with automated sample preparation
System
Column : Mastro C18 2.1*100mm, 3μm
Column Temp. : 50 ºC
Mobile Phase A : 0.1% Formic Acid - Water
Mobile Phase B : 0.1% Formic Acid - Methanol
Time Program : 5%B(0-4 min) – 100%B(4-5min) – 5%B(5min)
Flow Rate : 0.4mL/min
Injection Volume : 2μL
Ionization : ESI Positive
[LC] NexeraX2
[MS] LCMS-8040
Table 1 LC-MS/MS condition
Plasma spiked with four NOACs (Apixaban, Rivaroxaban,
Edoxaban, Dabigatran) were used for calibration. Six
calibration standards, QC samples and human plasma
samples were prepared. These were precipitated in the
CLAM-2000 using acetonitrile and vacuum ltration. In
contrast, manual sample preparation used a
centrifugation step following precipitation to remove the
protein content. The ltrated sample was injected into
LC-MS/MS.
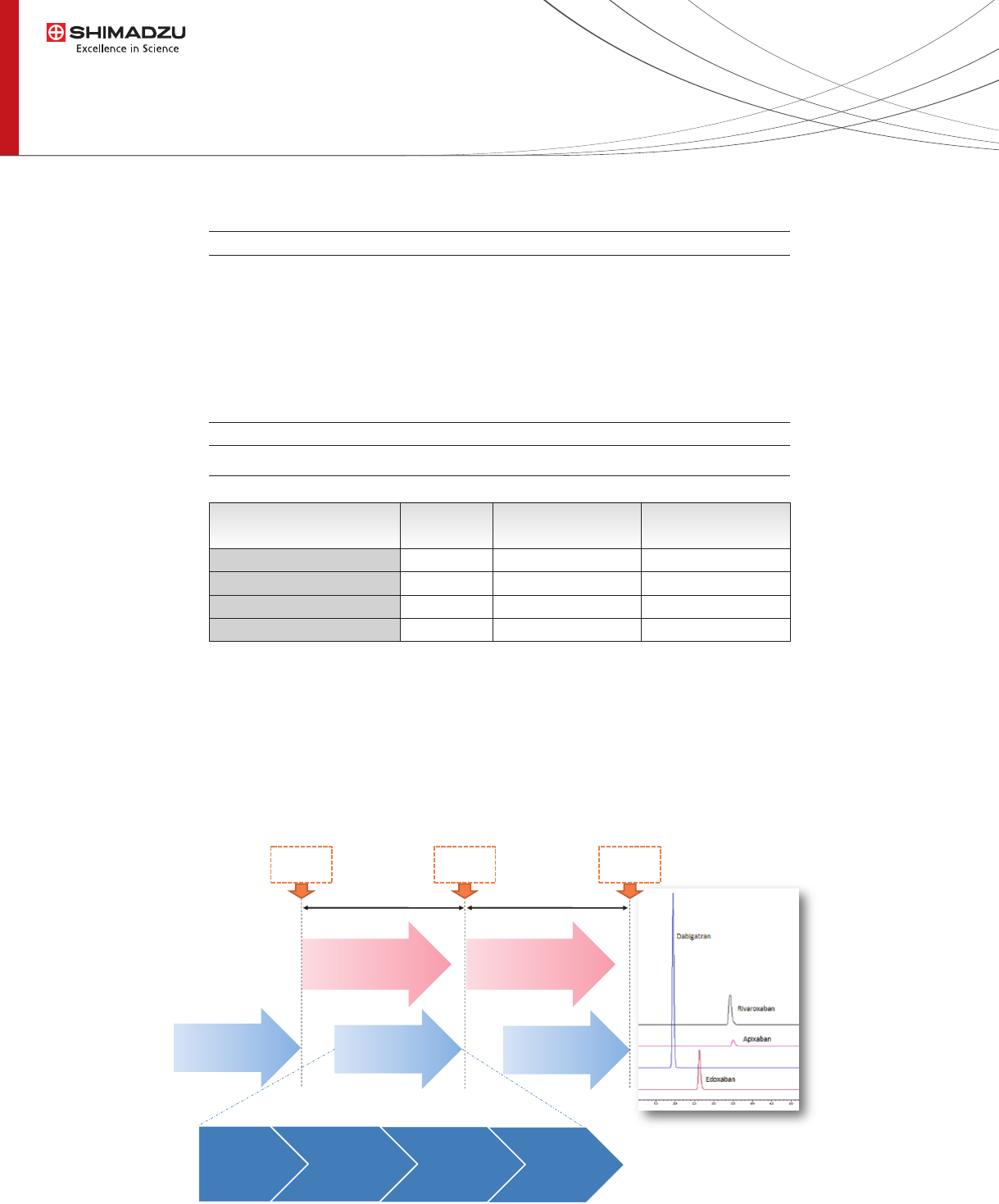
Sample preparation
Fig.2 Automated analysis from sample preparation to LC-MS/MS
460.20
436.10
548.20
472.00
+
+
+
+
Precursor (m/z)
443.10
145.00
152.20
289.05
Product (m/z)Polarity
Apixaban
Rivaroxaban
Edoxaban
Dabigatran
Compounds
MRM MS analysis
10min
Sample
Preparation
6min
Sample
Preparation
6min
MRM MS analysis
10min
Sample
Preparation
6min
12min 12min
Sample
injection
Filtration
• Time 120sec
Shaking
• Time 150sec
Reagent
Dispensing
• Acetonitrile
Sample
Dispensing
• Human plasma
Sample
injection
Sample
injection

4
High throughput analysis for novel oral anticoagulants using
LC-MS/MS system Integrated with automated sample preparation
Result and discussion
Each of the compounds were calibrated within the range
from 5ng/mL to 500ng/mL by the six calibration points
(5, 10, 20, 50, 200, 500ng/mL). Table 2 illustrates
linearity, accuracy and reproducibility of all compounds.
The calibration curves showed good linearity (R
2
>0.992).
The reproducibility (N=6) at six concentrations, including
LLOQ, of each compounds was excellent (CV<10%).
Different day reproducibility (N=6) for 5 days at three
concentrations (5, 50, 500ng/mL) as well (CV<15%).
Linearity, accuracy and reproducibility
Fig.3 Calibration curves for each compound
Table.2 Analytical performance
50
50
50
50
5
5
5
5
500
500
500
500
0.992
0.993
0.996
0.994
Concentrations of QC
samples (ng/mL)
LLOQ ULOQMiddle
4.95
5.84
2.92
2.19
9.16
4.65
2.52
0.85
1.53
2.21
1.26
1.05
% RSD (n=6)
LLOQ ULOQMiddle
10.44
7.20
5.49
5.54
13.74
12.30
8.64
7.28
14.23
6.20
5.61
6.22
% RSD (n=6)
LLOQ ULOQMiddle
r
2
Apixaban
Rivaroxaban
Edoxaban
Dabigatran
Compound
5 - 500
5 - 500
5 - 500
5 - 500
Range
(ng/mL)
99.1
107.8
109.1
111.4
95.7
104.8
100.0
101.7
107.1
106.0
99.2
96.9
Accuracy (%)
LLOQ ULOQMiddle
0 100 200 300 400
0
10000
20000
30000
40000
50000
60000
70000
80000
90000
2
3
4
5
6
7
0 100 200 300 400
0
25000
50000
75000
100000
125000
150000
175000
200000
2
3
4
5
6
7
Apixaban Rivaroxaban
0 100 200 300 400
0
50000
100000
150000
200000
250000
300000
350000
400000
450000
500000
550000
600000
650000
700000
750000
2
3
4
5
6
7
0 100 200 300 400
0
50000
100000
150000
200000
250000
300000
350000
400000
450000
500000
550000
600000
650000
2
3
4
5
6
7
Dabigatran
Edoxaban
Area
Concentration
Area
Concentration Concentration
Area
Area
Concentration