11_PO-CON1751E.pdf - 第3页
3 Quantitation of plasma metanephrine and normetanephrine by derivatization using an integrated LC-MS/MS analyzer equipped with fully-automated sample preparation device Fig. 2 Appearance of CLAM-LCMS system, and the pre…
2
Quantitation of plasma metanephrine and normetanephrine
by derivatization using an integrated LC-MS/MS analyzer equipped
with fully-automated sample preparation device
Introduction
• Although LC-MS/MS analysis manifests high repeatability
in measurement, overall reproducibility of an assay is
compromised by errors associated with manual sample
pretreatment. This also hinders standardization of assay
across multiple laboratories.
• Derivatization of is employed in LC-MS/MS analysis to
achieve better chromatographic separation or to enhance
favorable detection in MS. However, it involves series of
reagent addition and vortexing, making the pretreatment
prone to errors and hence low reproducibility.
• In this investigation, we evaluated tested whether or not
automated sample preparation device can be employed
to carry out complex sample pretreatment procedures,
such as required for chemical derivatization. For model
experiment plasma metanephrine and normetanephrine
was derivatized by reductive amination as shown in Fig. 1
to improve reversed phase column retention.
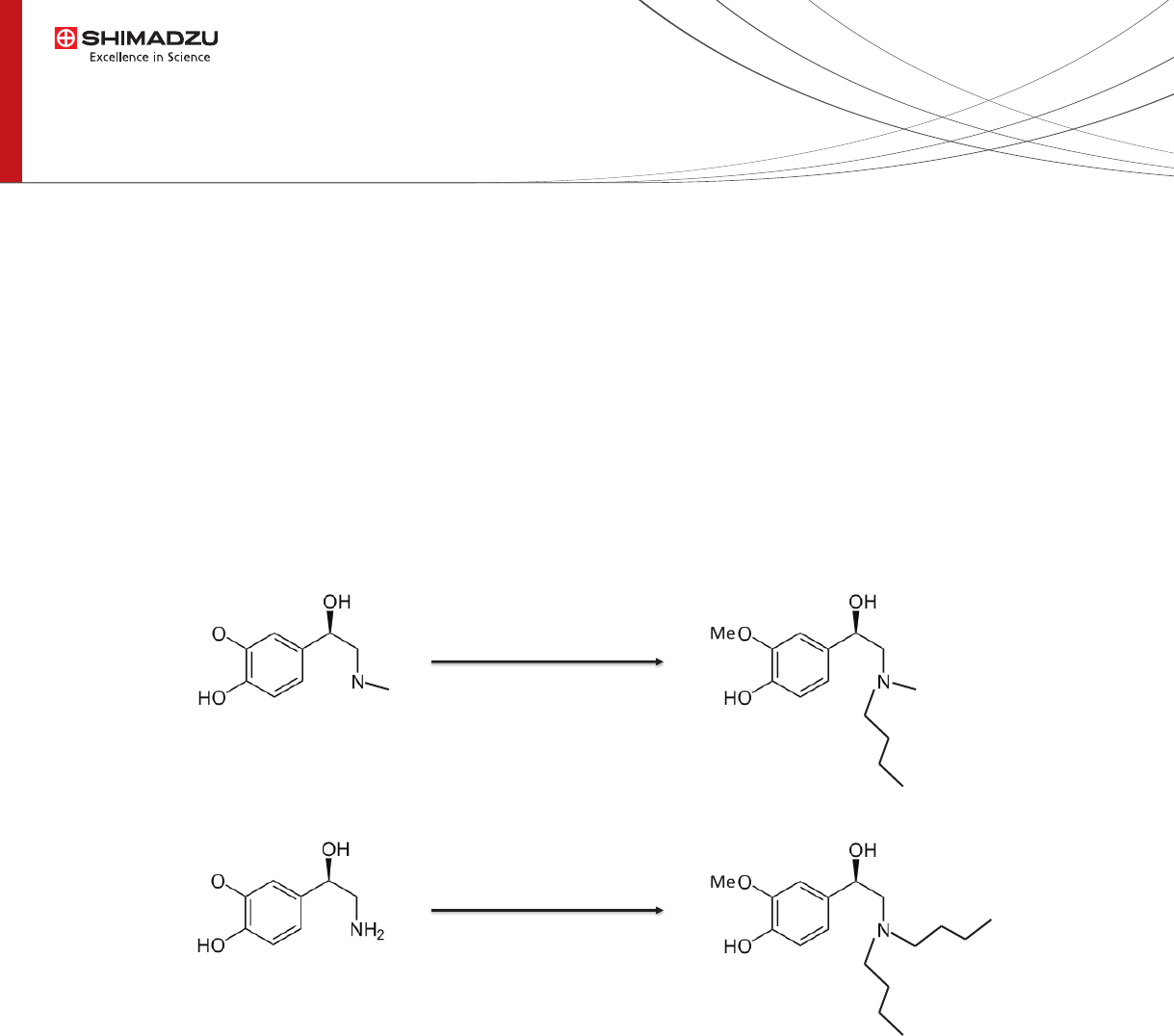
Fig. 1 Target compounds and their derivatization products obtained by reductive
amination using butanal as aldehyde and 2-picolineborane as reducing agent.
Me
Me
+ butanal
+ 2-picolineborane
+ acetic acid
+ butanal
+ 2-picolineborane
+ acetic acid
Dibutyl-NMN
Butyl-MN
Normetanephrine
(NMN)
Metanephrine
(MN)
3
Quantitation of plasma metanephrine and normetanephrine
by derivatization using an integrated LC-MS/MS analyzer equipped
with fully-automated sample preparation device
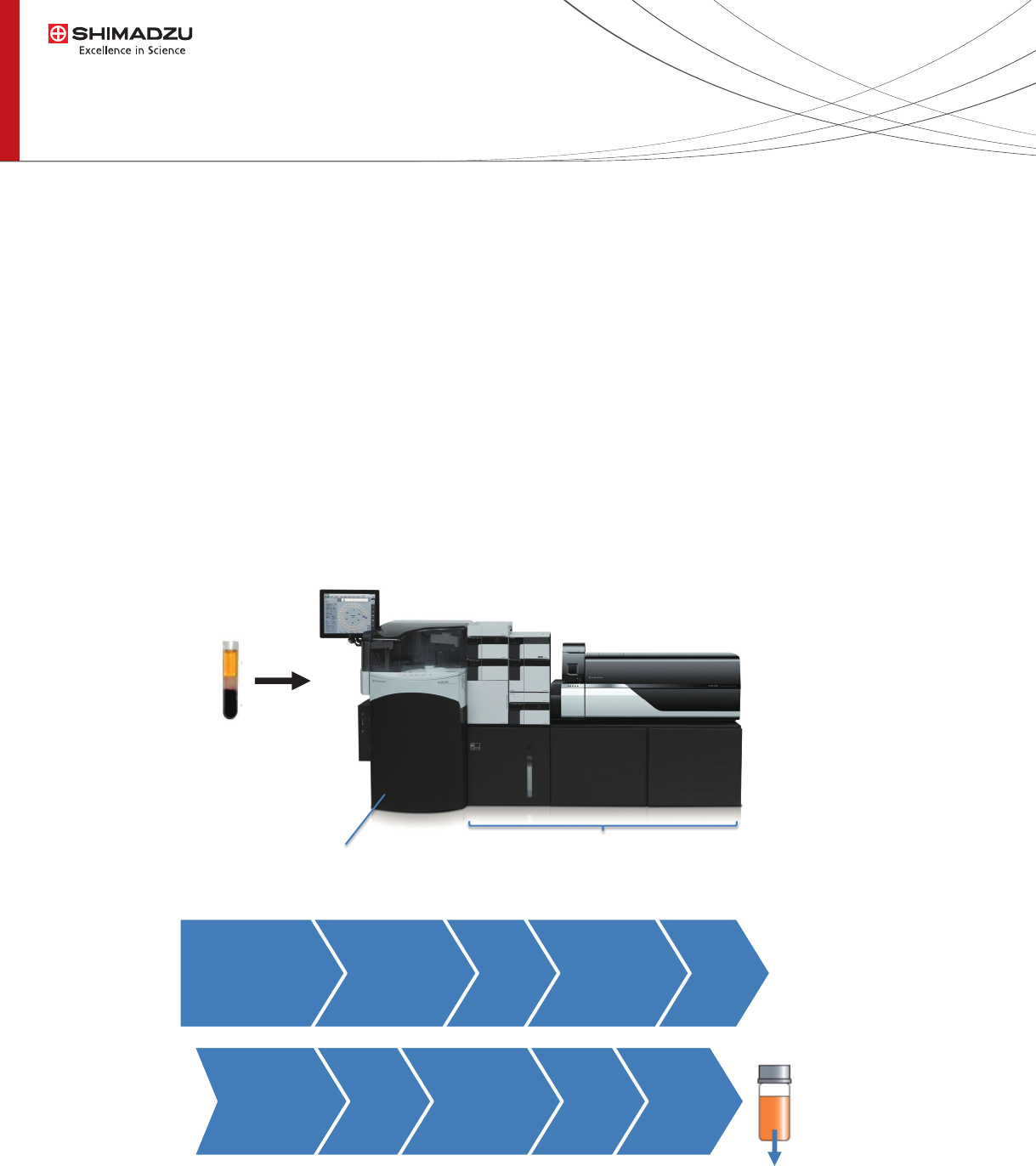
Fig. 2 Appearance of CLAM-LCMS system, and the pretreatement steps carried out for derivatization.
Fig. 2 illustrates the general appearance of the instrument setting, with CLAM-2000 connected to LCMS-8060 to
comprise an integrated “turnkey” type analyzer system. The pretreatment steps carried out are shown in the ow chart
that include 5 steps of reagent addition and 4 steps of vortexing. The procedure completes within 6 minutes, and it can
be ran parallelly with LC-MS/MS measurement so resulting in no wait-time between sequential analyses.
Automated sample preparation
Filtration
Vortex
120 sec
Vortex
60 sec
Plasma
sample
in collection
tube
CLAM-2000 LCMS-8060
Automatically
transferred to
autosampler for
seamless injection
s
e
c
Pretreatment steps (6 min)
Reagent
Dispensing
60 µL
Reagent1
Vortex
10 sec
IS
Dispensing
30 µL
IS
Sample
Dispensing
30 µL
plasma
Reagent
Dispensing
30 µL
Reagent2
Reagent
Dispensing
90 µL
Reagent3
Vortex
60 sec
Commercially available pooled plasma was used as sample matrix. IS solution (1 ppb of standard compounds and
deuterated internal standard in water), Reagent 1 (butanal/acetic acid = 25:75), Reagent 2 (7% 2-picolineborane in
EtOH, w/v) and Reagent 3 (5% aq. ammonia) were prepared in 6 mL glass container and placed in CLAM-2000 as
reagent reservoir.
Sample and reagents
Methods
4
Quantitation of plasma metanephrine and normetanephrine
by derivatization using an integrated LC-MS/MS analyzer equipped
with fully-automated sample preparation device
Metanephrine (MN)
MN-d3
Normetanephrine (NMN)
NMN-d3
254.15
257.15
296.20
299.20
Precursor m/z
(derivatized)
197.23
200.23
183.20
186.20
MW
-14
-21
-17
-21
CE (V)
236.15
154.10
278.20
154.10
Product m/zCompound
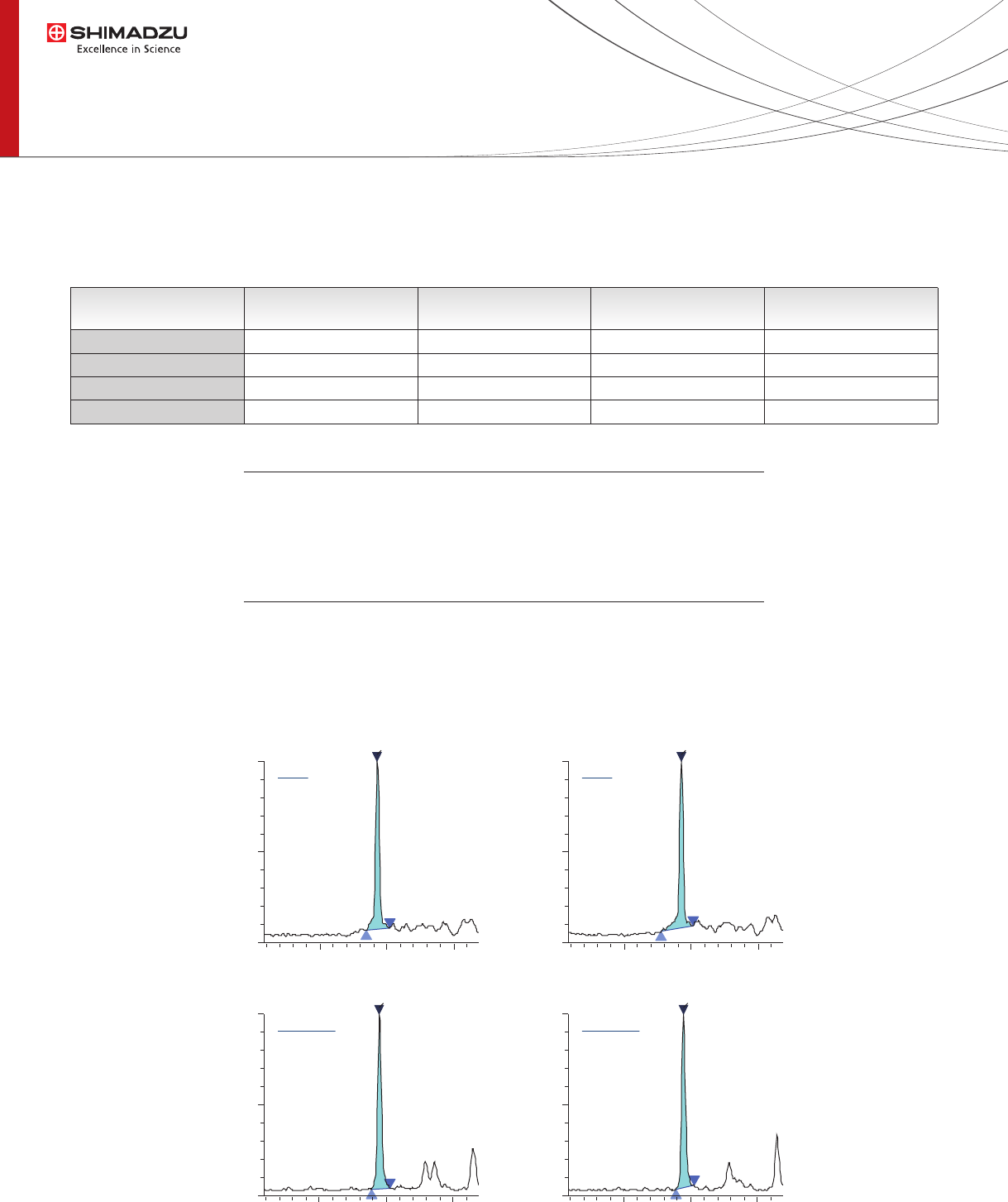
Fig. 3 MRM chromatograms of MN and MN-d3 spiked in control plasma at 1 ng/mL concentration.
Shown below are the list of target compounds, their MRM transitions and the HPLC condition for LC/MS/MS analysis.
Analytical conditions
Results
MN
(run 1)
MN
(run 2)
MN-d3
(run 1)
MN-d3
(run 2)
Column : Shimpack GISS C18 (100 mm x 2.0 mm, 3 μm)
Mobile phase A : 0.1% formic acid in water
Mobile phase B : Methanol
Flow rate : 0.4 mL/min
Column temp. : 40 °C
Injection volume : 1 μL
8.09e4ISTD 257.15>154.10
A=171010
H=80171
RT =2.446
2.0 2.5 3.0
0.00
%
100.00
8.34e4ISTD 257.15>154.10
A=171613
H=81989
RT =2.443
2.0 2.5 3.0
0.00
%
100.00
7.27e4Q 254.15>236.15
A=147336
H=68771
RT =2.426
2.0 2.5 3.0
0.00
%
100.00
8.02e4Q 254.15>236.15
A=158391
H=76320
RT =2.430
2.0 2.5 3.0
0.00
%
100.00