4_Application_News_No_C153.pdf - 第2页
Application News No. C153 Validation Test of the Fully Automated Pretreatment Analysis Method Calibration curves were created from the control blood plasma with standards added and the integrity of accuracy and precisi…

Application
News
No. C153
Liquid Chromatograph Mass Spectrometry
Simultaneous Analysis of Antiarrhythmic Drugs in
Human Blood Plasma Using the Fully Automated
Sample Preparation LC/MS/MS System
LAAN-A-LM-E123
During drug treatment with drugs that pose
administration management difficulties, such as drugs
with a narrow therapeutic range or drugs with a fine
line between toxicity and effectiveness, the blood
concentration of drugs in patients is measured to
determine the optimal dose and method of
administration for individuals based on
pharmacokinetic and pharmacodynamic analysis.
Application News No. C123 introduced an
investigation into optimizing the analysis workflow
including pretreatment by using the fully automated
sample preparation LC/MS/MS system that comprises
the CLAM-2000 fully automated LCMS sample
preparation unit and a high performance liquid
chromatograph mass spectrometer.
This article introduces a study which achieves a fast and
simultaneous analysis workflow of six antiarrhythmic
drugs with the fully automated sample preparation
LC/MS/MS system.
T. Tsukamoto, D. Kawakami
Analysis of Antiarrhythmic Drugs in Blood
Plasma with Fully Automated Pretreatment
Pretreatment of blood plasma samples for analysis
normally requires a process that involves
deproteinization by adding an organic solvent,
followed by centrifugal separation of solid
components and supernatant isolation. With the fully
automated sample preparation LC/MS/MS system,
these preparatory steps are done automatically just by
setting a blood collection tube after blood plasma
separation, and LC/MS/MS analysis is continuously
performed (Fig. 1). Pretreatment of the next sample can
also be performed in parallel with LC/MS/MS analysis,
which can greatly reduce the time required for each
sample analysis.
In this analysis example, a per-sample cycle time of 7
minutes was achieved from blood plasma
pretreatment to the simultaneous analysis of six
antiarrhythmic drugs and metabolites using LC/MS/MS
(Table 1 and Fig. 2).
Pretreatment Workflow of Blood Plasma Samples
Table 1 Antiarrhythmic Drugs and Metabolites
Compound
Molecular
Formula
MRM
Transition
m/z
Amiodarone C
25
H
29
I
2
NO
3
646.0 > 58.1
Desethylamiodarone
*
C
23
H
25
I
2
NO
3
618.0 > 72.1
Bepridil
C
24
H
34
N
2
O 367.1 > 84.1
Flecainide
C
17
H
20
F
6
N
2
O
3
415.0 > 301.0
Pilsicainide
C
17
H
24
N
2
O 272.9 > 110.1
Cibenzoline
C
18
H
18
N
2
262.9 > 115.0
Mexiletine C
11
H
17
NO
180.1 > 58.0
Mass Chromatograms of Human Blood Plasma
with Standard Additives
0.0 0.5 1.0 1.5 2.0 2.5 min
0.00
0.25
0.50
0.75
1.00
1.25
(x10,000,000)
1
2
3
5
4
6
7
1: Amiodarone
2: Desethylamiodarone
3: Bepridil
4: Flecainide
5: Pilsicainide
6: Cibenzoline
7: Mexiletine
Application
News
No.
C153
Validation Test of the Fully Automated
Pretreatment Analysis Method
Calibration curves were created from the control blood
plasma with standards added and the integrity of
accuracy and precision were evaluated based on the
analysis results of the QC samples (at concentrations of
n = 5) (Table 2). Good linearity was obtained in the set
concentration range for all antiarrhythmics. The
accuracy of the QC samples in the entire range,
including the quantitative lower limit, was within 100
± 15 %. Similarly, precision (%RSD) was within 15 % and
good repeatability was obtained.
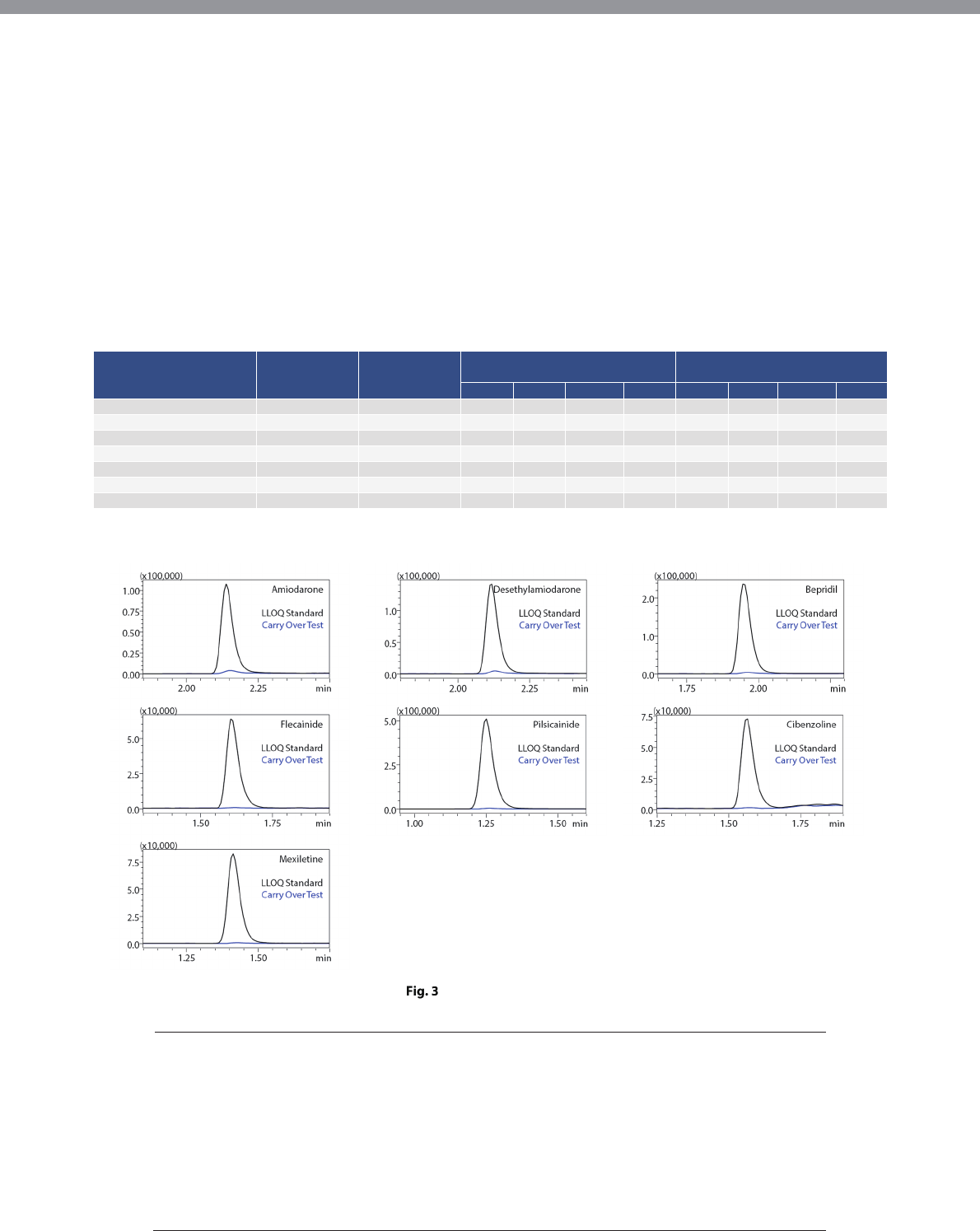
Immediately after analysis of the highest calibration
standard sample, blank blood plasma was measured to
check for carryover in the fully automated sample
preparation LC/MS/MS system. No significant carryover
was detected for any of the drugs upon comparison
with the peak intensity of the lowest calibration
standard sample (Fig. 3).
The above results show that the fully automated
sample preparation LC/MS/MS system used in this
article is capable of sufficiently reliable quantitative
analysis when performing consecutive analyses of
samples of wide-ranging concentrations.
Table 2 Validation Test Results for Simultaneous Analysis of Antiarrhythmic Drugs and Metabolites
Compounds
Cal. Range
[ng/mL]
Correlation
Coefficient
R
Accuracy
%
Precision
%RSD, n=5
LLOQ Low Medium High LLOQ Low Medium High
Amiodarone
*1
100-3000 0.9983 98.3 100.6 99.4 103.9 4.1 2.9 3.0 2.7
Desethylamiodarone
*1
100-3000 0.9987 99.2 98.9 101.1 100.3 5.3 4.2 3.6 4.2
Bepridil
*2
50-1500 0.9992 100.9 100.5 96.6 103.4 4.1 3.7 2.3 1.8
Flecainide
*2
50-1500 0.9987 98.1 98.7 96.7 101.4 4.7 3.3 2.4 2.4
Pilsicainide
*1
100-3000 0.9987 100.4 99.6 97.3 104.8 4.0 3.0 1.8 2.0
Cibenzoline
*2
50-1500 0.9987 102.4 101.4 99.1 102.9 4.2 3.4 3.0 2.4
Mexiletine
*1
100-3000 0.9984 104.5 107.4 106.3 107.8 3.8 3.9 2.6 2.6
*1: 100 ng/mL for LLOQ, 250 ng/mL for Low, 1000 ng/mL for Medium, 3000 ng/mL for High
*2: 50 ng/mL for LLOQ, 125 ng/mL for Low, 500 ng/mL for Medium, 1500 ng/mL for High
Carryover Test Results
Table 3 Analysis Conditions (Validation Test)
S
y
stem : CLAM-2000 + Nexera + LCMS-8060
Protocol : Plasma disp. 15 μL - acetonitrile disp. 285 μL - shaking at 1900 rpm, 120 sec - filtration for 90 sec
Column : Shimadzu GLC Mastro C18 (50 mmL. × 2.1 mmI.D., 3 μm)
Mobile Phase : A) 0.1% Formic acid - Water, B) 0.1% Formic acid - Methanol
Flow Rate : 0.4 mL/min
Time pro
g
ram : B Conc. 10% (0 min) – 100% (2 – 3.5 min) – 10% (3.51 – 6 min)
Column Temp. 50 °C In
j
ection Volume : 0.2 μL
Probe Volta
g
e : 2.0 kV (ESI-positive mode)
Interface Temp. : 300 °C DL Temp. : 250 °C
Block Heater Temp. : 400 °C Nebulizing Gas Flow : 3 L/min
Heatin
g
Gas Flow : 10 L/min Dr
y
in
g
Gas Flow : 10 L/min
Carry Over Test:
Analysis of Blank Plasma Following
The Highest Calibration Standard Sample
Application
News
No.
C153
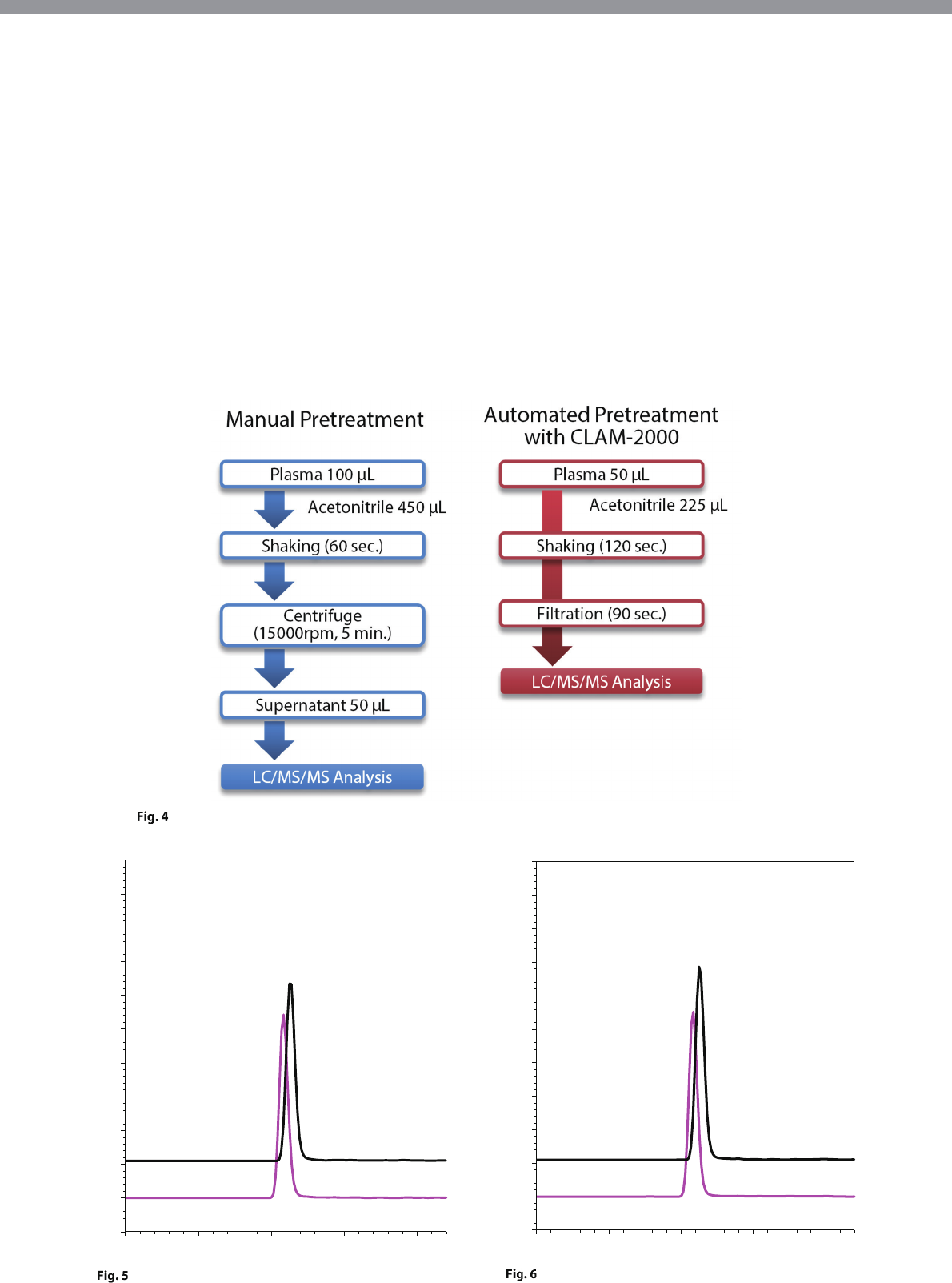
Comparative Test with Manual Pretreatment
A comparative test was performed between a manual
pretreatment method and the fully automated
pretreatment analysis method that employs the fully
automated sample preparation LC/MS/MS system.
Human blood plasma for measuring the concentration
of amiodarone was used.
The manual pretreatment method involved manually
isolating the blood plasma, adding acetonitrile, and
mixing to perform deproteinization. After centrifugal
separation of this sample, the supernatant was then
transferred to a vial for LC/MS/MS analysis. On the
other hand, the fully automated pretreatment analysis
method enabled the entire analysis process, from
blood plasma isolation to LC/MS/MS analysis, to be
performed completely automatically using the system
described in this article (Fig. 4).
A comparison of quantitative values between the
methods was performed for amiodarone and the
metabolite desethylamiodarone (Fig. 5 and 6, Table 4
and 5). In the wide range of concentrations detected
from the samples, there was favorable agreement
between the quantitative results of the manual
pretreatment method and the fully automated
pretreatment analysis method. The coefficient of
determination (R2) of both methods calculated from
these results was 0.95 or higher (Fig. 7 and 8).
The fully automated pretreatment analysis method
used by this system is a fast and low-burden analysis
technique that achieves quantitative results equivalent
to conventional manual pretreatment methods and we
anticipate its utilization into the future.
Pretreatment Workflow of the Manual Pretreatment Method and Fully Automated Pretreatment Analysis Method
Human Blood Plasma (Sample 3) Analysis Results Using
the Manual Pretreatment Method
Human Blood Plasma (Sample 3) Analysis Results Using
the Fully Automated Pretreatment Analysis Method
1.50 1.75 2.00 2.25 min
-0.1
0.0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1.0
(x100,000)
2:DEA 618.00>72.10(+) CE: -32.0
1:AMD 646.00>58.10(+) CE: -52.0
Manual
Pretreatment
Amiodarone
546 ng/mL
Desethylamiodarone
416 ng/mL
1.50 1.75 2.00 2.25 min
-0.1
0.0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1.0
(x100,000)
2:DEA 618.00>72.10(+) CE: -32.0
1:AMD 646.00>58.10(+) CE: -52.0
Automated Pretreatment
with CLAM-2000
Amiodarone
557 ng/mL
Desethylamiodarone
423 ng/mL