IPC-4552.pdf - 第14页
IPC-4552 Proposal June 2001 Nickel Thickness: Determini ng the require d thickne ss for the ni ckel barrie r layer was a rrived at easily through consensus. From t he input on “norm al” plat ing times bei ng used in the …
IPC-4552 Proposal June 2001
0
1
2
3
4
5
6
7
8
9
10
11
OEM PWB manf chem sup
p
group
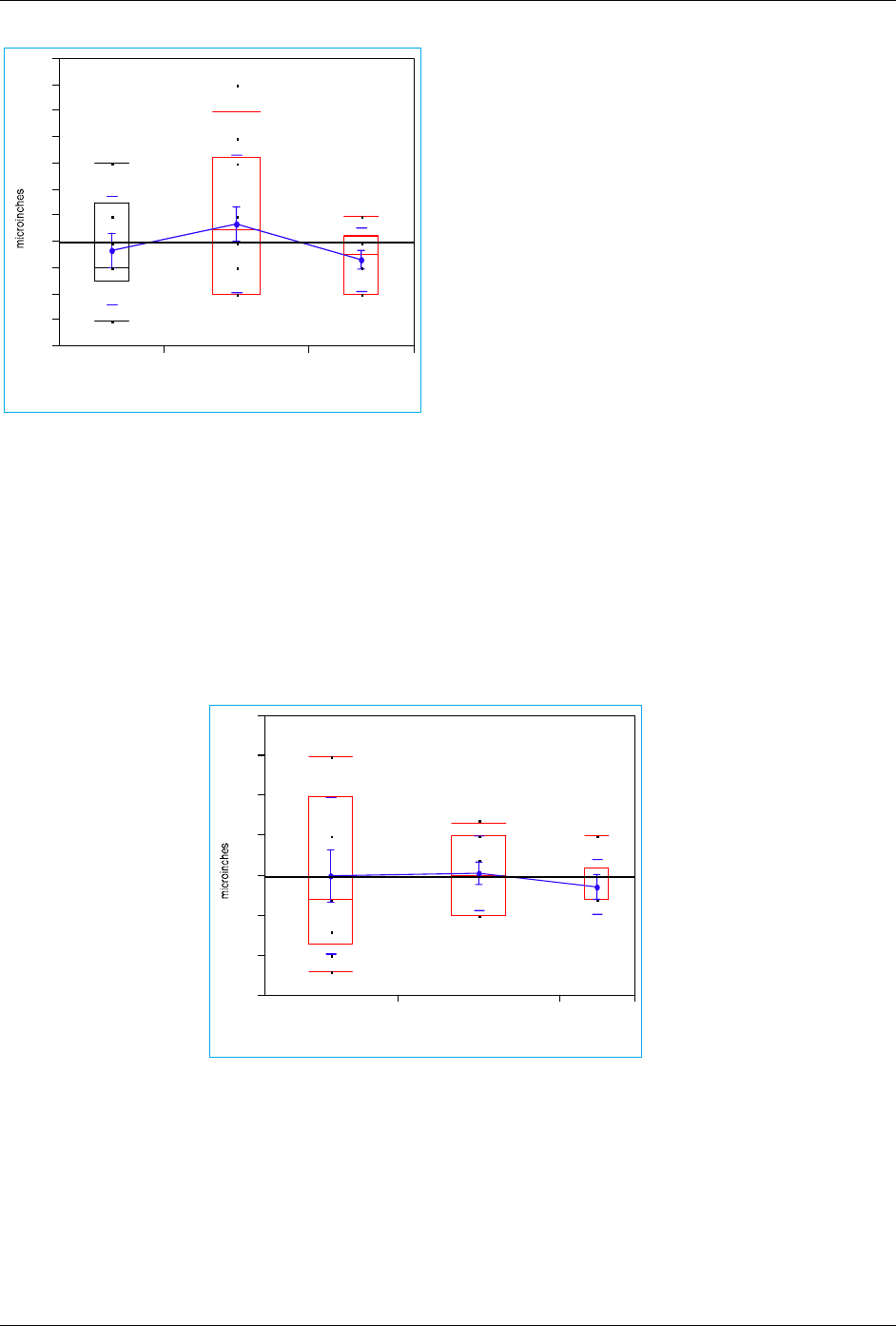
Means and Std Deviations
Level Number Mean Std Dev Std Err Mean
OEM 10 3.70000 2.11082 0.66750
PWB manf 14 4.71429 2.64367 0.70655
chem supplier 10 3.30000 1.25167 0.395
Fig 1: results from Gold thickness survey of 4552 members
0
50
100
150
200
250
300
350
OEM PWB manf chem su
p
group
Means and Std Deviations
Level Number Mean Std Dev Std Err Mean
OEM 9 150.000 99.1211 33.040
PWB manf 11 153.636 47.5968 14.351
chem supplier 5 136.000 35.7771 16.000
Fig 2: results from nickel thickness survey of 4552 members
Page 11 of 10
Download From http://bbs.infoeach.com
Download From http://bbs.infoeach.com

IPC-4552 Proposal June 2001
Nickel Thickness:
Determining the required thickness for the nickel barrier layer was arrived at easily through consensus. From the
input on “normal” plating times being used in the industry a specification range of 120 microinches to 240
microinches was agreed upon. At the 120 lower level it was felt this provided more than enough basis metal to act as
a diffusion barrier to copper migration as well as providing a suitable stable metal to solder to. The concern with
excessively thick nickel was not from a soldering viewpoint rather from the use of the finish for compliant pin and
the increase in insertion force required to overcome the reduction in plated through hole diameter.
Gold Thickness:
The immersion gold thickness was not that straight forward. A series of questions were raised:
• How much gold makes a good nickel solderability preservative?
• How much gold is needed for a minimum shelf life of I year?
• What are the processes out there capable of?
• Is better? Is more gold possible?
• How much gold is needed for “Contact Resistance”?
In an effort to answer these questions the committee designed and executed a series of capability studies. The studies
included:
• A capability study to determine what thickness of gold is the immersion process capable of.
• Solderability analysis by Wetting Balance method to determine the minimum gold thickness that is solderable.
• Solderability after aging.
• Contact Resistance testing using carbon membrane switches.
Round Robin Testing:
The committee has five chemical suppliers of ENIG, for the list of participants. What became apparent early in the
specification writing meetings was the variation in chemical management from the five different suppliers.
A quick round robin test was set up to determine the range of nickel and gold deposits that were achievable by the
five suppliers. The panels for the deposition rate test would also be used for solderability evaluations. All the panels
to be coated with ENIG were supplied from one PWB supplier and would be acid copper plated with no solder
mask. Based on the variations in chemical bath control etc, it was decided to level the playing field by having all the
nickel samples plated in baths at 1 MTO and all the gold baths at 25% of their useable life.
The test would consist of plating a maximum of 10 panels all at the same time in the nickel bath; some vendors
could only do 8 panels due to basket size constraints. They would all be plated at the same time to ensure correct
bath loading so that a statistically significant mean and standard deviation value could be obtained. For the gold
bath, a panel would be removed from the plating bath every two minutes until all of the panels were removed. Since
the gold bath is an immersion bath, the loading factor is not as much of an issue compared to the electroless nickel
bath.
Measuring Gold Thickness:
The ability to accurately measure the ENIG deposit could potentially be a paper/test method document in its own
rights. The thickness of the gold deposit poses some fundamental problems. A number of the committee members
have experienced measurement capability issues with their customers. These have ranged from using the wrong
calibration standards, i.e. using electrolytic Nickel electrolytic Gold standards
Page 12 of 10
Download From http://bbs.infoeach.com
Download From http://bbs.infoeach.com
IPC-4552 Proposal June 2001
0
1
2
3
4
5
6
SS XRF std XRF
XRF type
Means and Std Deviations
Level Number Mean Std Dev Std Err Mean
SS XRF 240 2.66542 1.24144 0.08013
std XRF 240 3.08750 1.47084 0.09494
Fig 3: comparison of Gold thickness values by XRF machine type
Fig 4: Wetting Balance coupon
FR4 0.062” thick, acid copper plated
6 X 3 mm SMD pads/side
Page 13 of 10
Download From http://bbs.infoeach.com
Download From http://bbs.infoeach.com