IPC-CM-770D-1996.pdf - 第146页
IPC-CM-770 Januaty 1996 no deleterious impact on reliability and do not need to be removed from the assembly with a post solder defluxing operation. Other no clean fluxes, no residue, are formulated with no rosin and uti…

January
1996
IPC-CM-770
All gold plated leads and wires that are hand soldered or
surface mounted should be pretinned or solder dipped to
remove the gold prior to component soldering. Care should
be exercised to not adversely affect the component ele-
ments during this treatment.
27.2.1.1 Cleaning Prior to Tinning
Component leads
can be cleaned with a mechanical cleaning tool that does
not impart damage to the component or the component
lead. Other methods of cleaning can be used, provided that
these methods do not damage the component. Knives,
emery cloth, sandpaper, steel wool and other abrasive
should not be used.
27.2.2 Substrate Preparation
Printed wiring boards
should be tested for solderability before assembly.
If
sol-
derability is not acceptable, then pretreatment prior to
assembly may be required to enhance solderability and
subsequent quality of the soldered assembly. Solder mask-
ing may or may not be present dependent on specifications.
Typical pretreatments are degreasing (surface contamina-
tion, e.g., dirt, oils), brightening (chemical activation of
solder and/or copper), and baking (moisture). Baking of the
substrate may be necessary in order to prevent delamina-
tion of the substrate.
27.3 Materials
All soldering processes are only capable
of achieving optimum process yields if the materials used
in those processes are not substandard. Regardless of the
particular soldering process used (dip, wave, reflow), all
solder processes follow the same basic steps: flux, preheat,
and soldering. Some newer technologies use other materi-
als such as adhesives either in conjunction with or to
replace soldering. Adhesive attachment of components is
particularly attractive with temperature sensitive devices,
and for securing surface mount devices. The various types
of alternative solder materials will be discussed here.
27.3.1 Flux
Flux must have properties such that it
(1)
chemically removes the surface oxide or tarnish and
(2)
keeps the surface clean until the solder has melted and
flowed over the fluxed surface.
Soldering fluxes have been divided into three general cat-
egories. The traditional flux specifications classify fluxes
on the basis of their chemical make-up or flux base (rosin
base fluxes, for example, are classified as R, RMA or
RA).
J-STD-004 utilizes a unified approach to flux classification
based on fundamental, intrinsic corrosive and conductive
properties of flux and flux residues, rather than specifying
the flux base.
Flux are specified according to one of the following three
types per J-STD-004:
L
=
Low or no flux/flux residue activity
M= Moderate flux/flux residue activity
H= High flux/flux residue activity
Inorganic fluxes are not permitted for electronics soldering.
The flux and the cleaning process (or lack thereof) are
directly interdependent.
27.3.1.1
Many common fluxes use natural rosin as a
base. This natural product, derived from the gum of pine
trees, is a mix of abietic acid and numerous dehydrogena-
tion products.
Rosin is a glassy, non-crystalline mixture of organic acids
which are inert up to their softening point and only assume
an acidic nature when molten. After melting and resolidifi-
cation the hard glassy properties render the residue once
again inert. Residues from other added chemical activator
compounds usually become encapsulated in the rosin resi-
due, which renders them non- corrosive. The effectiveness
of this safeguard, however, depends on the quantity and
nature of the activator used.
The acidity of pure rosin alone is usually insufficient to
clean surfaces to be soldered,
so
rosin fluxes are usually
enhanced by a variety of chemicals called activators. Com-
mon activators are inorganic halides, organic halides, car-
boxylic acids, amines and halogenated amines. The degree
of activation achieved by the various chemicals depends
upon the compound used and the quantity. Since the over-
all activity developed is often a synergistic product of more
than one activator, activation is usually quantified not by
formulation alone but rather by some secondary property
such as the ability of the residue extract to dissolve a cop-
per from the “Copper Mirror Test”-or by its ionic con-
ductivity. The level of activator affects the rate of wetting.
27.3.1.2 Organic Acid (Water Washable) Fluxes
These
fluxes are significantly more active and aggressive than
rosin fluxes in removing oxides from the surfaces to be
soldered. They use strong organic acids and salts to achieve
these properties. As such they are more forgiving of poor
solderability characteristics of the surfaces to be soldered.
The increased activity yields flux residues which are more
corrosive than the residues of rosin fluxes. Because of their
characteristics it is necessary to completely remove the
residue with a post soldering cleaning operation to prevent
early failure of the soldered assembly.
27.3.1.3 No Clean Fluxes
(Low
ResiduelNo Residue).
This family of fluxes includes both rosin or modified resin
fluxes and non- rosin fluxes. The activators used are gener-
ally weak organic dicarboxcylic acids.
The low solids rosidresin based fluxes leave small
amounts of residue after soldering and except where these
may interfere with bed of nails testing or other post solder-
ing operations or operating characteristics generally have
6-9
COPYRIGHT Association Connecting Electronics Industries
Licensed by Information Handling Services
COPYRIGHT Association Connecting Electronics Industries
Licensed by Information Handling Services

IPC-CM-770 Januaty 1996
no deleterious impact on reliability and do not need to be
removed from the assembly with a post solder defluxing
operation.
Other no clean fluxes, no residue, are formulated with no
rosin and utilize a mixture of one or more weak dicarbox-
cylic acids and wetting agents to provide the activation
needed to enhance soldering. These fluxes leave little or no
residue and do not need to be removed from the assembly
with a post solder defluxing operation.
An inert gas atmosphere, such as nitrogen, is often used to
enhance the soldering process when using these more
benign fluxes.
27.3.2 Solders
Solders are generally metal alloys with
melting points in the range -150°C to -400°C. Below this
temperature range, alloys are commonly called fusible
alloys: above this they are called brazes. Tin-lead alloys are
most common, although more complex compositions had
been developed for special applications.
For electrical soldering, alloys near the eutectic composi-
tion (63% tin 37% lead) have the required combination of
properties. Although compositions either side of the eutec-
tic have higher liquidus (completely melted) temperatures
desirable for higher ambient temperature applications,
remember that the initial melting point (at which solder
softens) is 183"C, the same for all tin-lead alloys with
compositions between 20% and
98%
tin.
The range of properties of tin-lead alloys can be varied by
adding other metals such as bismuth or indium to lower the
melting point, or antimony, silver, etc. to increase hardness
and fatigue resistance. Alloys containing less than
10%
tin
are used for applications involving temperatures below
-40°C.
27.3.2.1 Solder Alloy Selection
Selecting a solder alloy
for a particular assembly depends on the expected operat-
ing conditions and on the types of components used. For
example, the mechanical and fatigue properties of solder
may be more important for surface mounted components
than through hole components.
With respect to temperature, consider the initial melting
point of the alloy when seeking an alloy for high temper-
ature applications or for multiple step soldering applica-
tions. Table 27-1 is a list of the most common solder alloys
and their melting temperatures.
27.3.2.2 Solder Purity
As soldering processes become
progressively more automated, final product quality
depends increasingly on consistent material quality and in
particular on solder purity. Solder should be purchased to a
standard of purity, as specified in J-STD-006.
Even very pure solder will pick up impurities from the
substrates, component leads, jigs and even the solder bath
Table 27-1 Solder Alloys
Alloy Melting Temperature
("C)
63/37 tin/lead
.....................................................................
183
60/40 tin/lead
..............................................................
183-1 91
60/40 lead/indium
.......................................................
195-225
52/48 indiumkin
..................................................................
11 8
62/36/2 tin/lead/silver
.........................................................
179
96/4 tin/silver
......................................................................
221
95/5 tin antimony
........................................................
235-240
10/88/2 tin/lead/silver
..................................................
268-290
1/97.5/1.5 tin/lead silver
.....................................................
309
43/43/14 tin/lead bismuth
...........................................
144-1 63
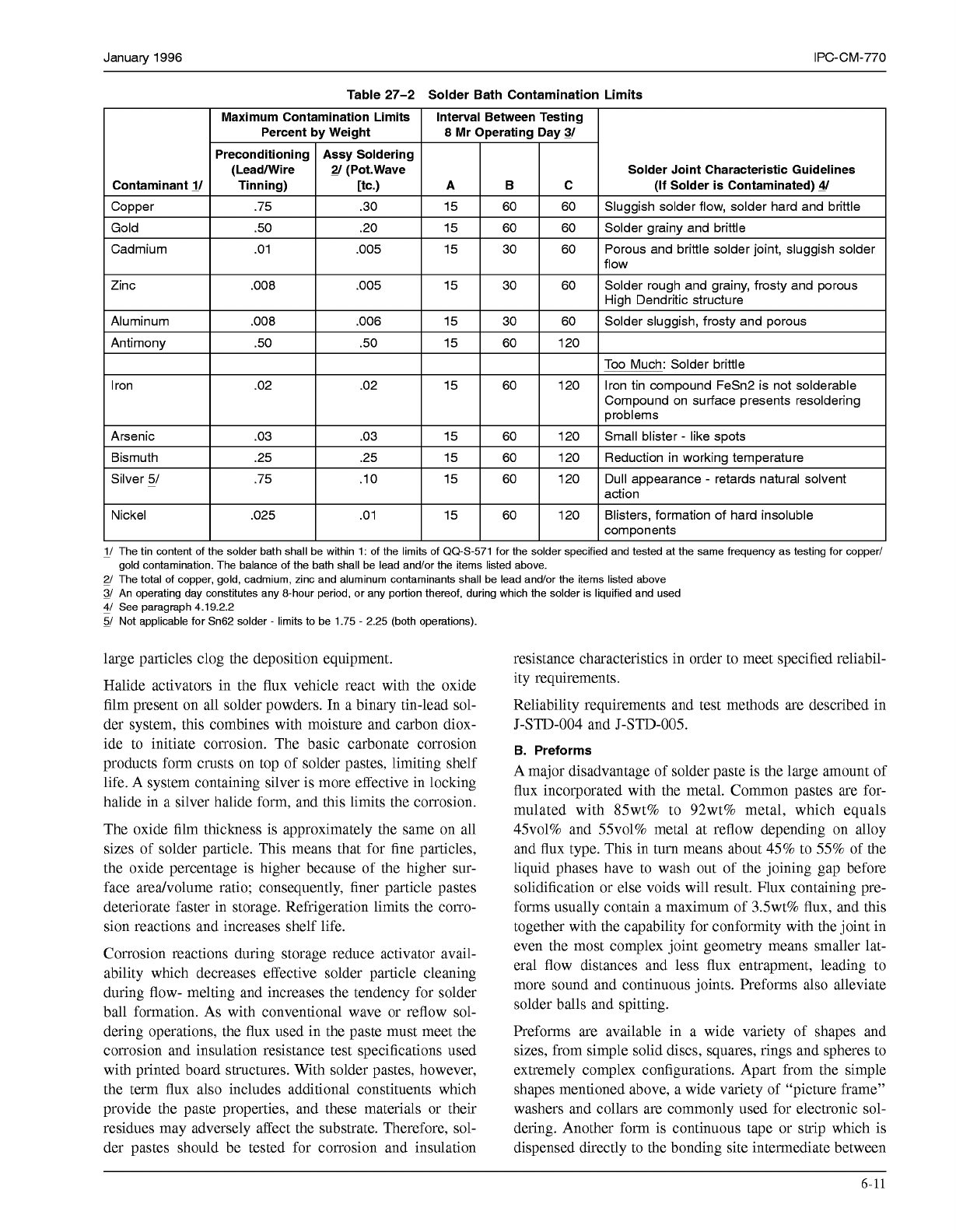
and machine tracks themselves. This limits the time life of
a solder bath. Typical limits for various likely impurity ele-
ments and the defects these can cause are also shown in
Table 27-2. Tin content should be tested with the same
frequency as testing for copper/gold contamination. The
balance of the path is lead and/or the items listed in Table
27-2.
27.3.2.3 Solder Forms
Various forms of solder can be
used to join components and printed board structure lands.
Solder can be supplied in ingot, wire, platings, preforms,
and pastes.
The solder form used in a particular soldering operation is
dependent on the method of heat application, component
placement methodology, and solder alloy selected.
A. Solder Pastes
Solder pastes are rarely used for soldering through board
mounted components, but they are used extensively for
surface mount where the variety of application methods
lends variety and flexibility to component mount and place-
ment schemes. These pastes can be applied to printed board
structures using three basic methods: screen printing, sten-
ciling and dispensing.
The properties of the solder paste affect the application
results. Solder pastes consist of a fluhinder and solder
alloy particles. The rheology of the fluxhinder keeps the
heavy solder particles
(85
to
89%
by weight) evenly dis-
persed under normal temperature and pressure conditions.
The fluhinder contains pure rosin or synthetic resins, acti-
vators, solvents, lubricants and thickeners. The activator
removes surface oxides and tarnishes during the soldering
process. The solvents adjust the solder paste to proper vis-
cosity. Since the solvents alone do not provide adequate
body to support the metal powder, thickeners are added.
They also give the solder paste the proper texture and life-
time. Solder particle shape and size also significantly influ-
ence deposition characteristics. Well rounded particles flow
more readily through small openings and give better pat-
tem definition, while the irregularly shaped particles or
6-10
COPYRIGHT Association Connecting Electronics Industries
Licensed by Information Handling Services
COPYRIGHT Association Connecting Electronics Industries
Licensed by Information Handling Services
January
1996
IPC-CM-770
Table
27-2
Solder Bath Contamination Limits
Solder Joint Characteristic Guidelines
1/ The tin content of the solder bath shall be within 1: of the limits of 00-5-571 for the solder specified and tested at the same frequency as testing for copper/
2/ The total of copper, gold, cadmium, zinc and aluminum contaminants shall be lead and/or the items listed above
3/
An operating day constitutes any 8-hour period, or any portion thereof, during which the solder is liquified and used
gold contamination. The balance of the bath shall be lead and/or the items listed above.
&/
See paragraph 4.19.2.2
5/ Not applicable for Sn62 solder
-
limits to be 1.75
-
2.25 (both operations).
large particles clog the deposition equipment.
Halide activators in the flux vehicle react with the oxide
film present on all solder powders. In a binary tin-lead sol-
der system, this combines with moisture and carbon diox-
ide to initiate corrosion. The basic carbonate corrosion
products form crusts on top of solder pastes, limiting shelf
life. A system containing silver is more effective in locking
halide in a silver halide form, and this limits the corrosion.
The oxide film thickness is approximately the same on all
sizes of solder particle. This means that for fine particles,
the oxide percentage is higher because of the higher sur-
face aredvolume ratio; consequently, finer particle pastes
deteriorate faster in storage. Refrigeration limits the corro-
sion reactions and increases shelf life.
Corrosion reactions during storage reduce activator avail-
ability which decreases effective solder particle cleaning
during flow- melting and increases the tendency for solder
ball formation. As with conventional wave or reflow sol-
dering operations, the flux used in the paste must meet the
corrosion and insulation resistance test specifications used
with printed board structures. With solder pastes, however,
the term flux also includes additional constituents which
provide the paste properties, and these materials or their
residues may adversely affect the substrate. Therefore, sol-
der pastes should be tested for corrosion and insulation
resistance characteristics in order to meet specified reliabil-
ity requirements.
Reliability requirements and test methods are described in
J-STD-O04 and J-STD-005.
B. Preforms
A major disadvantage of solder paste is the large amount of
flux incorporated with the metal. Common pastes are for-
mulated with 85wt% to 92wt% metal, which equals
45~01% and 55~01% metal at reflow depending on alloy
and flux type. This in turn means about 45% to
55%
of the
liquid phases have to wash out of the joining gap before
solidification or else voids will result. Flux containing pre-
forms usually contain a maximum of 3.5wt% flux, and this
together with the capability for conformity with the joint in
even the most complex joint geometry means smaller lat-
eral flow distances and less flux entrapment, leading to
more sound and continuous joints. Preforms also alleviate
solder balls and spitting.
Preforms are available in a wide variety of shapes and
sizes, from simple solid discs, squares, rings and spheres to
extremely complex configurations. Apart from the simple
shapes mentioned above, a wide variety of “picture frame”
washers and collars are commonly used for electronic sol-
dering. Another form is continuous tape or strip which is
dispensed directly to the bonding site intermediate between
6-11
COPYRIGHT Association Connecting Electronics Industries
Licensed by Information Handling Services
COPYRIGHT Association Connecting Electronics Industries
Licensed by Information Handling Services