IPC-TM-650 EN 2022 试验方法.pdf - 第143页
1 Scope This test method specifies two methods for the determination of the acid value of a flux of type L, M or H. Method A is a potentiometric titration method and is to be considered the reference method. Method B is …
T
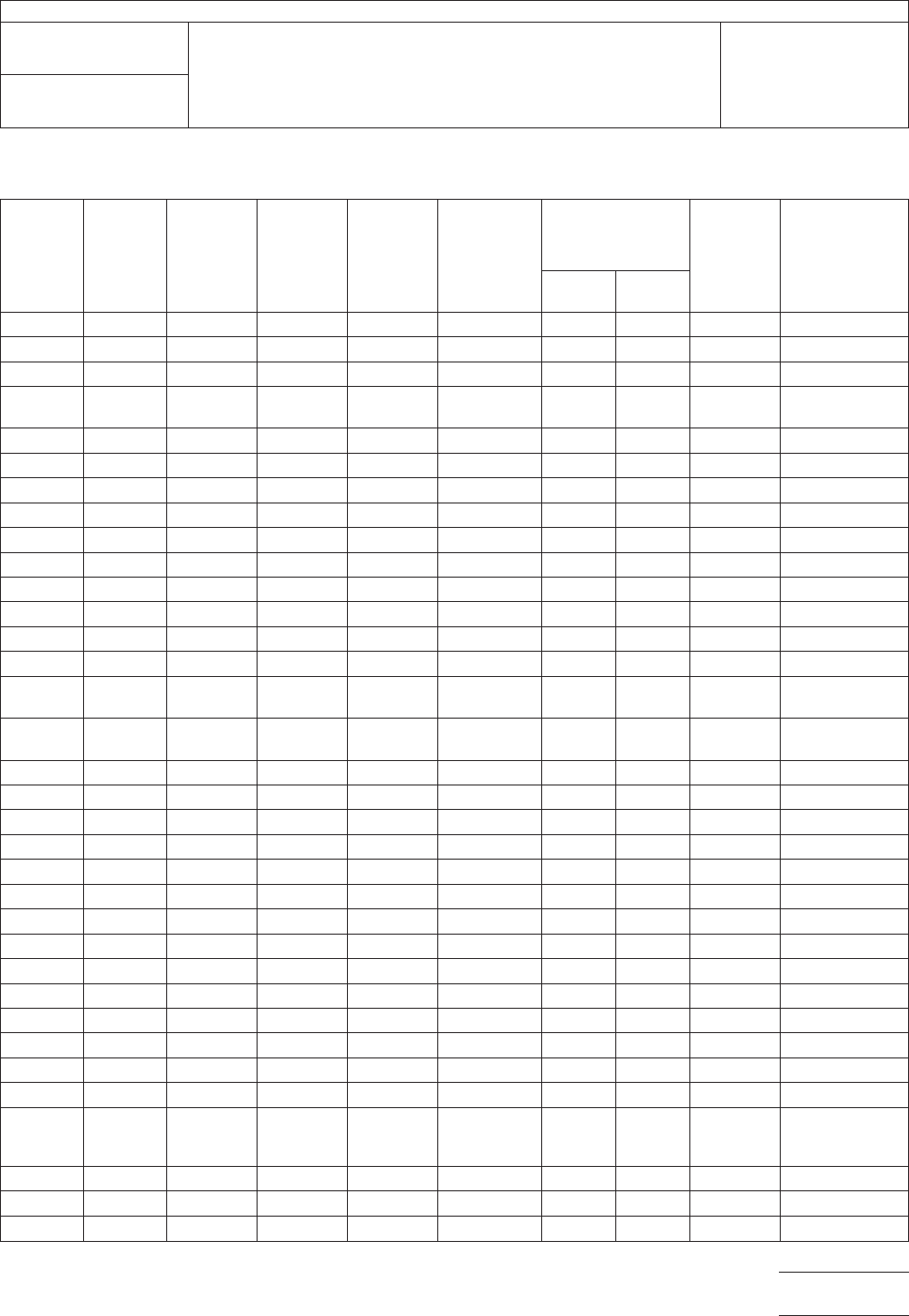
able 1
Cloth
No.
Nominal
W
idth
Inches
Thickness
Inch
Weight
Per
Square
Yard
Ounces
Yarn,
Warp,
and Fill Construction
Minimum
Breaking
Strength Per
Inch of Width
Yards per
Standard
Roll Type of Weave
Warp
Pounds
Fill
Pounds
106
38 0.0015 0.85 900-1/0 56x56 46 52 250 ± 25 Plain
108 38 0.0020 1.43 900-1/2 60x47 70 40 250 ± 25 Plain
112 38 0.0030 2.09 450-1/2 40x39 82 80 250 ± 25 Plain
113 38 0.0030 2.46 450-1/2
900-1/2
60x64 123 60 250 ± 25 Plain
116 38 0.0040 3.16 450-1/2 60x58 123 120 250 ± 25 Plain
119 38 0.0040 2.80 450-1/2 54x50 110 100 250 ± 25 Plain
120 38 0.0040 3.16 450-1/2 60x58 125 120 250 ± 25 4-harness satin
126-150 38 0.0060 5.50 150-1/2 34x32 225 195 250 ± 25 Plain
126 38 0.0065 5.37 450-3/2 34x32 205 185 250 ± 25 Plain
127 38 0.0070 6.00 450-3/2 42x32 225 185 250 ± 25 Plain
128 38 0.0070 6.00 225-1/3 42x32 250 200 250 ± 25 Plain
128-150 38 0.0067 6.00 150-1/2 42x32 250 200 250 ± 25 Plain
140 38 0.0100 8.70 450-4/3 32x21 400 290 125 ± 25 Plain
141 38 0.0100 8.70 225-2/3 32x21 400 290 125 ± 25 Plain
143 38 0.0090 8.90 225-3/2
450-1/2
49x30 611 56 125 ± 25 4-harness satin
143-150 38 0.0086 9.40 150-2/2
450-1/2
49x30 660 70 125 ± 25 4-harness satin
148 38 0.0120 10.10 450-3/5 30x19 450 360 125 ± 25 Plain
149 38 0.0120 10.80 225-2/4 30x19 450 360 125 ± 25 Plain
161 38 0.0150 12.20 450-4/5 28x16 550 450 125 ± 25 Plain
162 38 0.0150 12.20 225-2/5 28x16 450 350 125 ± 25 Plain
164 38 0.0150 12.60 225-4/3 20x18 500 450 125 ± 25 Plain
164-150 38 0.0140 13.00 150-4/2 20x18 500 450 125 ± 25 Plain
181 38 0.0085 8.90 225-1/3 57x54 340 330 125 ± 25 8-harness satin
181-150 38 0.0080 9.50 150-1/2 56x54 350 325 125 ± 25 Plain
182 38 0.0130 12.40 225-2/2 60x56 440 400 125 ± 25 8-harness satin
182-150 38 0.0134 12.65 150-1/3 60x56 440 400 125 ± 25 Plain
183 38 0.0180 16.75 225-3/2 54x48 650 620 75 ± 15 8-harness satin
184 38 0.0270 25.90 225-4/3 42x36 950 800 50 ± 10 8-harness satin
184-150 38 0.0246 24.16 150-4/2 44x35 950 800 50 ± 10
573-150 38 0.0110 8.00 150-3/2 16x16 335 316 125 ± 25 Plain
1000-150 38
and
44
0.0130 9.66 150-4/2 16x14 450 410 125 ± 25 Plain
1044-150 38 0.0220 19.20 ...... 16x14 450 410 125 ± 25 Plain
1523-150 38 0.0140 11.70 150-3/2 28x20 525 400 125 ± 25 Plain
1527-150 38 0.0150 12.90 150-3/3 17x17 535 485 125 ± 25 Plain
IPC-TM-650
Number
2.3.11
Subject
Glass
Fabric Construction
Date
4/73
Revision
P
age3of3
电子技术应用 www.ChinaAET.com

1
Scope
This
test method specifies two methods for the
determination of the acid value of a flux of type L, M or H.
Method A is a potentiometric titration method and is to be
considered the reference method.
Method B is an alternative, visual end-point, titration method.
2
Applicable Documents
ISO 9455
Soft
Soldering Fluxes, Test Methods
IPC-TM-650
Test
Methods Manual
2.3.34 Solids Content, Flux
IPC
J-STD-004
Requirements
for Soldering Fluxes
3
Test Specimen
A
minimum of 2.0 grams of liquid flux, 10
grams of solder paste, 150 grams of cored wire or 10 grams
of solder preforms.
4
Apparatus and Reagents
4.1 General
4.1.1
Use
only reagents of recognized analytical quality and
only deionized water.
4.1.2
Ordinary
laboratory apparatus.
4.2
For Potentiometric Titration Method (Method A)
4.2.1
Tetrabutyl
ammonium hydroxide, 0.1 M (0.1 mole/L).
Use a commercially available standard solution or one pre-
pared from a commercially available concentrated standard
solution by dilution with 2-propanol (4.2.2). Alternatively, pre-
pare a 0.1 M tetrabutyl ammonium hydroxide solution by dilut-
ing a commercial concentrated solution with 2-propanol and
standardize this solution against an accurately weighed
amount of benzoic acid (about 0.5 g) dissolved in dimethylfor-
mamide, previously neutralized to thymol blue.
4.2.2
2-Propanol.
Neutralized with tetrabutyl ammonium
hydroxide solution (4.2.1) to a faint pink color using phenol-
phthalein as an indicator.
4.2.3
Ethanol,
anhydrous. Neutralized with tetrabutyl ammo-
nium hydroxide solution (4.2.1) to a faint pink color using phe-
nolphthalein as an indicator.
4.2.4 Toluene.
Neutralized with tetrabutyl ammonium
hydroxide solution (4.2.1) to a faint pink color using phenol-
phthalein as an indicator.
4.2.5
Ethanol/toluene
mixture. Mix equal volumes of the
neutralized anhydrous ethanol (4.2.3) and neutralized toluene
(4.2.4).
4.2.6
Phenolphthalein
Indicator.
4.2.7 Millivoltmeter
or pH meter.
4.2.8
Glass
electrode.
4.2.9
Saturated
calomel, or silver chloride/silver, electrode.
4.2.10
Magnetic
or mechanical stirrer with variable speed
drive.
4.2.11 Burette
capable of delivering aliquots of 0.1 ml to 1.0
ml.
4.3
For Titration with Visual End-Point (Method B)
4.3.1
Ethanol,
anhydrous. Neutralized with potassium
hydroxide, 0.1 M in alcohol (4.3.5), to a faint pink color using
phenolphthalein as an indicator.
4.3.2
Toluene.
Neutralized with potassium hydroxide, 0.1 M
in alcohol (4.3.5), to a faint pink color using phenolphthalein as
an indicator.
4.3.3
Ethanol/toluene
mixture. Mix equal volumes of the
neutralized anhydrous ethanol (4.3.1) and neutralized toluene
(4.3.2).
4.3.4 2-Propanol.
Neutralized with potassium hydroxide, 0.1
M in alcohol (4.3.5), to a faint pink color using phenolphthalein
as an indicator.
2215
Sanders Road
Northbrook, IL 60062-6135
IPC-TM-650
TEST
METHODS MANUAL
Number
2.3.13
Subject
Determination
of Acid Value of Liquid Solder Flux -
Potentiometric and Visual Titration Methods
Date
06/04
Revision
A
Originating Task Group
Flux Specifications Task Group (5-24a)
Material
in this Test Methods Manual was voluntarily established by Technical Committees of IPC. This material is advisory only
and its use or adaptation is entirely voluntary. IPC disclaims all liability of any kind as to the use, application, or adaptation of this
material. Users are also wholly responsible for protecting themselves against all claims or liabilities for patent infringement.
Equipment referenced is for the convenience of the user and does not imply endorsement by IPC.
P
age1of3
ASSOCIA
TION CONNECTING
ELECTRONICS INDUSTRIES
®
电子技术应用 www.ChinaAET.com

4.3.5
Potassium
hydroxide solution, 0.1 M in alcohol. Use a
commercially available standard solution or one prepared from
a commercially available concentrated standard solution by
dilution with ethanol (4.3.1). Alternatively, prepare an approxi-
mate 0.1 M potassium hydroxide solution by dissolving 3 ±
0.1 g potassium hydroxide (KOH) in 500 ml of ethanol (4.3.1),
and standardize this solution against an accurately weighed
amount of benzoic acid (about 0.5 g) dissolved in ethanol
(4.3.1).
4.3.6
Phenolphthalein
indicator solution. Add 1g of phenol-
phthalein to approximately 50 ml methanol and mix. When
dissolved, dilute to 100 ml with methanol and mix.
5
Procedures
5.1 Potentiometric Titration (Method A)
5.1.1
By
preliminary experiments, determine whether the
sample is soluble in 2-propanol, anhydrous ethanol, toluene or
the ethanol/toluene mixture. If the sample is not completely
soluble in any of these solvents, select the one in which the
sample appears to be the most soluble. If the sample is
equally soluble in all four solvents, then use neutralized
2-propanol (4.2.2).
5.1.2
Carry
out the following procedure, in triplicate, on the
flux sample.
5.1.2.1
Weigh, to the nearest 0.001 g, 2 to5goftheliquid
flux sample, taking steps to prevent loss of volatile matter
during the weighing. The larger size (≈5 g) sample is required
for very low solids fluxes. Transfer the weighed sample to a
250 ml low form beaker.
5.1.2.2 Dilute
the sample to 100 ml with 2-propanol (4.2.2),
or the selected solvent (4.2.3 to 4.2.5), according to the solu-
bility characteristics of the flux. Cover with a watch glass and
dissolve the flux by gentle agitation.
5.1.2.3
Place
the beaker on the stand of the titration assem-
bly with the electrodes, stirrer and burette in position. Adjust
the speed of the stirrer to give vigorous stirring without splash-
ing. Titrate with the 0.1 M tetrabutyl ammonium hydroxide
solution (4.2.1), adding 1.0 ml portions and recording the pH
or mV meter reading after each addition. As the end-point is
approached, reduce the additions of titrant to 0.1 ml and con-
tinue titrating past the end-point.
5.1.2.4
Plot
the pH or potential values against the volume of
titrant added to obtain the titration curve. The point of inflec-
tion of the curve corresponds to the end-point of the titration.
Note: The point of inflection of the curve may conveniently be
determined by using the derivative curve.
5.1.2.5
Carry out a blank determination, using reagents
only, for comparison purposes.
5.2
Visual Titration (Method B)
5.2.1
By
preliminary experiments, determine whether the
sample is soluble in 2-propanol, anhydrous ethanol, toluene or
the ethanol/toluene mixture. If the sample is not completely
soluble in any of these solvents, select the one in which the
sample appears to be the most soluble. If the sample is
equally soluble in all four solvents, then use neutralized etha-
nol (4.3.1) as the selected solvent.
5.2.2
Carry
out the following procedure, in triplicate, on the
flux sample.
5.2.2.1
Weigh,
to the nearest 0.001 g, sufficient flux sample
to correspond to approximately1gofnonvolatile matter (see
IPC-TM-650, Test Method 2.3.34), taking steps in the case of
liquid flux samples to prevent loss of volatile matter during the
weighing.
5.2.2.2
Transfer
the weighed sample to a suitable flask or
beaker and add 100 ml of the selected solvent. Stir until the
sample has dissolved as completely as possible. Do not heat.
5.2.2.3
Add
three drops of phenolphthalein indicator solu-
tion (4.3.6) and titrate with the 0.1 M potassium hydroxide
solution (4.3.5) until a faint pink color persists throughout the
titrated solution for 15 seconds.
5.2.2.4
Carry
out a blank determination, using reagents
only, for comparison purposes.
5.3
Calculation of Results
The
acid value is expressed in
milligrams of potassium hydroxide per gram of nonvolatile
matter, regardless of the alkali used to perform the titration.
5.3.1
The
acid value (expressed in milligrams of potassium
hydroxide per gram of nonvolatile matter) is given by:
56.11VM
mS
IPC-TM-650
Number
2.3.13
Subject
Determination
of Acid Value of Liquid Solder Flux -
Potentiometric and Visual Titration Methods
Date
06/04
Revision
A
P
age2of3
电子技术应用 www.ChinaAET.com