IPC-TM-650 EN 2022 试验方法.pdf - 第216页
Leave the flask at room temperature for 30 min ± 2 min after combustion. The generated gas is absorbed into the alkali solution to produce Cl - /Br - ion solution. Transfer the solution from the combustion flask to a vol…

1 Scope The purpose of this test method is to measure the
amount of chlorine and bromine compounds in base materi-
als. This test method is applicable to reinforced base materi-
als with a minimum thickness of 0.3 mm [0.012 in] and to
un-reinforced base materials with a minimum thickness of
0.08 mm [0.0031 in].
A combustion flask is used to extract ionic and covalent halo-
gen from the sample, and Ion Exchange Chromatography is
used for the quantitative analysis of halogen content.
2 Applicable Documents All terms and definitions in this
document conform to IPC-T-50,
Terms and Definitions for
Interconnecting and Packaging Electronic Circuitry.
The IPC Test Method is generated and written, verbatim, from
the IEC 61189-2 TEST 2C12: Total Halogen Content in
Base Materials.
3 Test Specimens Rigid or flexible base materials shall be
used for the test provided they meet the minimum thickness
requirements described in the scope. The copper foil (if appli-
cable) shall be removed from the test specimens by etching
by any industry acceptable etching method or by mechanical
peeling before test.
For reinforced base materials, the number of test specimens
shall not be less than five (5) with a minimum size of 1 cm x
1 cm [0.39 in x 0.39 in]. For unreinforced base materials, the
number of test specimens shall not be less than five (5) with a
minimum size of 1 cmx1cm[0.39 in x 0.39 in].
Wash the specimens thoroughly in distilled or deionized water.
For reinforced base materials, dry the sample at 105 °C [221
°F] +5/- 0 °C [+9 °F/-0 °F] for one hour + 0.25 hour /-0 hour.
For unreinforced base materials, wipe off the water with a lint-
free cloth or paper wiper and leave them to dry at room tem-
perature and atmospheric pressure for a minimum of one
hour.
4 Apparatus & Materials
a. Ion exchange chromatograph with a detection limit of 10
ppm or better.
b. Analytical balance with an accuracy of approximately 1
mg in weight or better.
c. Knife.
d. Tweezers.
e. Vinyl gloves.
f. Lint-free cloth, paper wipers or equivalent.
g. Quantitative filter paper.
h. Combustion flask, or equivalent.
i. Oxygen (99.9 % purity or better).
j. Gas pressure regulator.
k. Flint striker, or another ignition device.
l. Wash bottle.
m. Beaker.
n. Micro-pipette.
o. Flasks (various sizes).
p. Potentiometric titrator for silver nitrate, if necessary.
q. Platinum basket for holding sample.
r. Alkali solution.
s. Ethyl alcohol.
t. Chlorine ion standard solution for chromatograph.
u. Bromine ion standard solution for chromatograph.
v. Silver nitrate titrant, if potentiometric titration using silver
nitrate is used.
5 Procedure
5.1 Combustion Procedure
Weigh the specimen using
the analytical balance and record the result.
Place approximately 50 ml alkali solution in a combustion flask
to act as an absorbent of combustion gas.
Fill the combustion flask with oxygen.
Insert a test specimen into the combustion flask as shown in
Figure 1.
Insert a piece of filter paper into the combustion flask as
shown in Figure 1 to act as a fuse/flame starter.
Apply several drops of ethyl alcohol on the test specimen for
improving the ignition of the specimen and then ignite the
specimen.
3000 Lakeside Drive, Suite 309S
Bannockburn, IL 60015-1219
IPC-TM-650
TEST METHODS MANUAL
Number
2.3.41
Subject
Test Method for Total Halogen Content in Base
Materials
Date
04/06
Revision
Originating Task Group
Halogen Free Materials Subcommittee (4-33)
Material in this Test Methods Manual was voluntarily established by Technical Committees of IPC. This material is advisory only
and its use or adaptation is entirely voluntary. IPC disclaims all liability of any kind as to the use, application, or adaptation of this
material. Users are also wholly responsible for protecting themselves against all claims or liabilities for patent infringement.
Equipment referenced is for the convenience of the user and does not imply endorsement by IPC.
Page1of3
ASSOCIATION CONNECTING
ELECTRONICS INDUSTRIES
®
Leave the flask at room temperature for 30 min ± 2 min after
combustion. The generated gas is absorbed into the alkali
solution to produce Cl
-
/Br
-
ion solution.
Transfer the solution from the combustion flask to a volumet-
ric flask. Introduce deionized water in the flask until the total
amount is 100 ml of the test solution.
Remove the suspended subjects from the test solution by fil-
tration or centrifugation if necessary.
Follow the same procedure outlined above without the test
specimen to prepare a reference test solution (blank) without
combustion of a test sample.
Note: Chlorine and bromine contents shall be measured
beforehand without any test specimen in the combustion
flask.
5.2 Chemical Analysis Inject the test fluid gathered from
the flask in 5.1 into the inlet of an ion exchange chromato-
graph shown in Figure 2.
Analyze the peak area/height of the Cl
-
/Br
-
conductivity on the
recorder and obtain Cl
-
/Br
-
concentration from a calibration
curve.
Obtain Cl
-
/Br
-
ion concentrations of the reference by compar-
ing its conductivity using the standard solution. The test
solution is compared to the reference solution (blank). Any
contaminant in the reference solution will be subtracted from
the test solution.
Titrate test solution using silver nitrate if concentration of Cl
-
/
Br
-
ions is higher than 1 wt%. An example of analyzing condi-
tions for the ion exchange chromatography is given in Table 1.
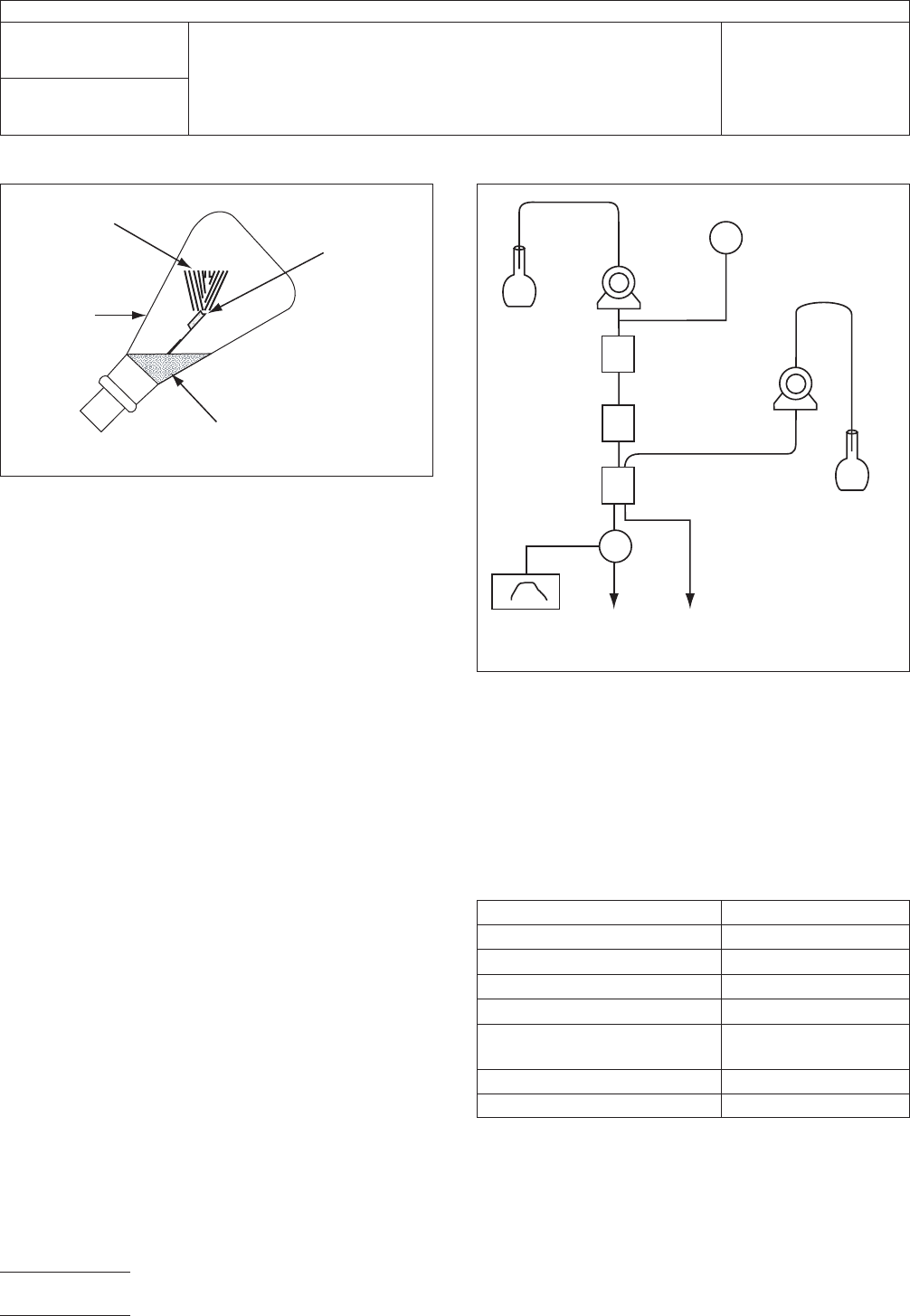
IPC-2341-1
Figure 1 Absorb Combustion Gas Using a Combustion
Flask
Alkali solution
as absorbent
Platinum basket
(Test piece, filter
as fuse)
Fire
Combustion
flask (or
equivalents)
IPC-2341-2
Figure 2 Composition of Ion Exchange Chromatograph
Recorder
Waste
Fluid
Waste
Fluid
Conductivity
Detector
Suppressor
Separation
Column
Precolumn
Elimination Fluid
Delivering Pump
Elimination
Fluid
Inlet
Eluant
Eluant
Delivering
Pump
Table 1 Example of Analyzing Conditions
for the Ion Exchange Chromatography
Item/Material Definition/Quantity
Eluant Alkali solution
Eluant delivering rate 1.0 ml/min to 2.0 ml/min
Elimination fluid H
2
SO
4
Elimination fluid and delivering rate 1.0 ml/min to 2.0 ml/min
Column Precolumn,
separation column
Suppressor Suppression for anion
Detector Conductivity meter
IPC-TM-650
Number
2.3.41
Subject
Test Method for Total Halogen Content in Base Materials
Date
04/06
Revision
Page2of3

Analyzing conditions depends on the test devices, test speci-
mens, their composition and environment.
5.3 Calculation of Halogen Content Insert the concentra-
tion of halogen ion (Cl
-
/Br
-
) obtained in 5.2 in the following for-
mula to obtain halogen contents in the specimens:
Chlorine (wt%) =
{[Cl
-
concentration in the test fluid (ppm) x
Cl
-
Dilution ratio in the fluid] -
[Cl
-
concentration in the reference (ppm) x
Cl
-
Dilution ratio in the reference]} x
{quantity of test solution (ml) /
mass of the test specimen} x 10
-7
Note: For the bromine content, use the same equation but
insert Br
-
values in the place of Cl
-
.
Note: The dilution ratio is the amount of added water as com-
pared to the total amount of solution in the test flask. If the
amounts used were 50 ml each, the dilution ratio would be 2.
6 Report In addition to the general requirements for report-
ing, the report shall include:
a. Test method number and revision.
b. Date of the test.
c. Identification and description of the specimen.
d. Average chloride content of the five (5) specimens in ppm.
e. Average bromide content of the five (5) specimens in ppm.
f. Average total halogen content of the five (5) specimens in
ppm.
g. Any deviation from this test method.
h. Date of the test.
i. Name of the person conducting the test.
IPC-TM-650
Number
2.3.41
Subject
Test Method for Total Halogen Content in Base Materials
Date
04/06
Revision
Page3of3